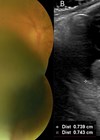
With the 2006 Collaborative Ocular Melanoma Study Group (COMS) study demonstrating no difference in survival outcomes for participants randomised to either iodine-125 brachytherapy or enucleation, the majority of primary uveal melanomas in the United States are now treated with brachytherapy. However, practices differ on dosimetry, planning methodology, and prescription doses. This retrospective study utilised data collected from nine institutions in the US between 2010-2014 and a survey conducted in 2018 querying current treatment and prescription methods. Data collected included: tumour dimensions, tumour class, plaque size and location, radiation dose, and practice patterns relating to imaging and planning. Participant eligibility was based on: a preoperative clinical diagnosis of uveal melanoma, gene expression profile confirmation, and minimum of three months follow-up. Amongst the 406 participants included for analysis there was an even distribution between males and females as well as between the affected and fellow eye. The mean age at plaque insertion was 60 years. Tumour characteristics were detailed as: mean 7.8mm basal diameter, 4.6mm mean apical height, majority class one tumour, and commonest location being between the macula and equator. It was reported that notched plaques, compared to non-notched plaques, provided a larger clinical margin, with a mean margin of 3.3mm ±1.3 and 2.7mm ±0.9 respectively (p<0.0001). Regarding dose, six institutions utilised a dosage of ≥85Gy for the apex, whilst the other two delivered doses <85Gy. Doses to the optic disc, macula and lens were consistent between institutions except one for optic disc and two for lens. Image based planning by eight institutions was via Eye Physics Plaque Simulator software which is not Food & Drug Administration (FDA) approved, however, at present no other software corrects for the key parameters such as carrier placement or plaque material. Study limitations included: number of centres responding, self-reported data, US-only data. Furthermore, data may be biased due to excluding participants with no gene expression profile, an iris tumour, or a tumour too small for biopsy. Future studies would help curb uncertainties on minimum effective dose, margins for control, and investigate practices outside of the US.
Practice patterns of uveal melanoma management with Iodine-125 brachytherapy
Reviewed by Annes Ahmeidat
Practice patterns for the treatment of uveal melanoma with iodine-125 plaque brachytherapy: ocular oncology study consortium report 5.
CONTRIBUTOR
Annes Ahmeidat
MBChB, BSc, Ninewells Hospital and Medical School, Dundee, UK.
View Full Profile