Medico-Legal
Issues with standard practice: Luer lock syringes in cataract surgery
For a few years, we have worked together with the practice variance surveys to tease out differences in the way we work and to discuss issues that arise. I hope that we have learnt some lessons and reflected on what...
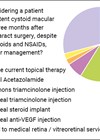
High-stakes scenarios in retinal vein occlusion: A survey of medico-legal implications
Central retinal vein occlusion (CRVO) is the second most common retinal vascular disease after diabetic retinopathy, and its clinical complexity often intersects with medico-legal scrutiny [1]. Missteps in timely diagnosis, inadequate follow-up or poor documentation can lead to patient harm...
The results of the last survey Jun25
The first question refers to whether we check blood glucose levels prior to cataract surgery. Almost three quarters of you do which was interesting. We know that poor glycaemic control (manifesting as a raised HbA1c) has implications for wound healing...
The results of the last survey Apr25
Postoperative endophthalmitis is arguably the worse possible complication of cataract surgery. We are a victim of our own success as the procedure has such an amazing safety profile that when things go wrong patients are bound to feel aggrieved and...
The results of the last survey Feb25
*Please be aware that this data does not form part of a peer reviewed research study. The information therein should not be relied upon for clinical purposes but instead used as a guide for clinical practice and reflection. The sample...
The results of the last survey Dec24
This edition’s survey results are fascinating. They focus more on your impressions and views rather than on the facts of your clinical practice. Effectively I asked you to take the role of an expert and comment on the cases I...
The results of the last survey Oct24
*Please be aware that this data does not form part of a peer reviewed research study. The information therein should not be relied upon for clinical purposes but instead used as a guide for clinical practice and reflection. The sample...
The results of the last survey Aug24
In the first case we were faced with a 55-year-old patient with narrow, potentially occludable drainage angles, no significant cataract, and persistently raised intraocular pressures (IOPs) of 28mmHg with early disc compromise, and normal vision in each eye. It is...
The results of the last survey Jun24
When I was in my training and even in my early years as a consultant, I did not fully understand the difference between different lenses. When asked my preference of hydrophilic versus hydrophobic intraocular lenses (IOLs) I really did not...
The results of the last survey Apr24
It has been a few years now since we started these surveys and I continue to be amazed by the variance in our practice. As a patient, I think I would expect there to be more consistency and evidence-based practice....
The results of the last survey Feb24
Thank you to all those who participated in this edition’s survey and for those of you who attended my recent Medico-legal Seminar at the Royal College of Ophthalmologists. It was a fascinating and educational day, made a success by the...
The results of the last survey Dec23
*Please be aware that this data does not form part of a peer reviewed research study. The information therein should not be relied upon for clinical purposes but instead used as a guide for clinical practice and reflection. Hopefully by...