This case report describes the management of a giant pigment epithelial detachment (PED) in neovascular age-related macular degeneration (nAMD) using half-dose aflibercept.
An 80-year-old woman presented with a huge PED (maximum height 1338µm) and multiple risk factors for retinal pigment epithelium (RPE) tear. Despite dose reduction, a grade 3 RPE tear developed two weeks after treatment, although anatomical improvement and partial visual recovery were observed.
This case highlights that even half-dose anti-vascular endothelial growth factor (anti-VEGF) therapy carries a substantial risk of RPE tear in high-risk PEDs. Careful risk assessment and thorough patient counselling are essential when considering treatment in such cases.
Anti-vascular endothelial growth factor agents have transformed the management of nAMD, enabling disease stabilisation and reducing the risk of severe visual loss. However, RPE tears remain a recognised complication of PED in nAMD, occurring either spontaneously or following anti-VEGF treatment. These tears can lead to substantial, often irreversible visual decline. Key risk factors include considerable PED height and diameter, a jagged RPE surface and hyperreflective spaces beneath the RPE.
Multimodal imaging has significantly enhanced the understanding of RPE tear development and has become essential in identifying high-risk eyes, as well as guiding treatment strategies. Imaging also plays a central role in patient counselling, particularly given the lack of alternative therapeutic options to prevent RPE tears in large PEDs. Previous reports have demonstrated successful use of reduced-dose anti-VEGF therapy for managing large PEDs, suggesting a potential role in lowering contractional forces exerted on the RPE.
We present a rare case of a giant vascularised PED treated with intravitreal half-dose aflibercept. This case is notable for its considerable PED height (maximum 1338µm), jagged nasal RPE, and recent rapid enlargement – features strongly associated with RPE tear risk. The patient received a half-dose aflibercept injection and was closely monitored for progression.
Case report
An 80-year-old Caucasian woman was being monitored in our clinic for a macular PED in her right eye. Her ocular history included uncomplicated bilateral cataract extraction with intraocular lens implantation one year prior. Her medical history included well-controlled diabetes mellitus and hyperlipidaemia.
Over three months, the PED progressively increased in size, accompanied by worsening visual symptoms. The decision was made to initiate anti-VEGF therapy. At baseline, best-corrected logMAR visual acuity (BCVA) measured 0.9 (pinhole 0.76) in the right eye and 0.06 in the left. Anterior segment examination was unremarkable. Fundus examination showed a large PED in the right eye and macular reticular drusen in the left.
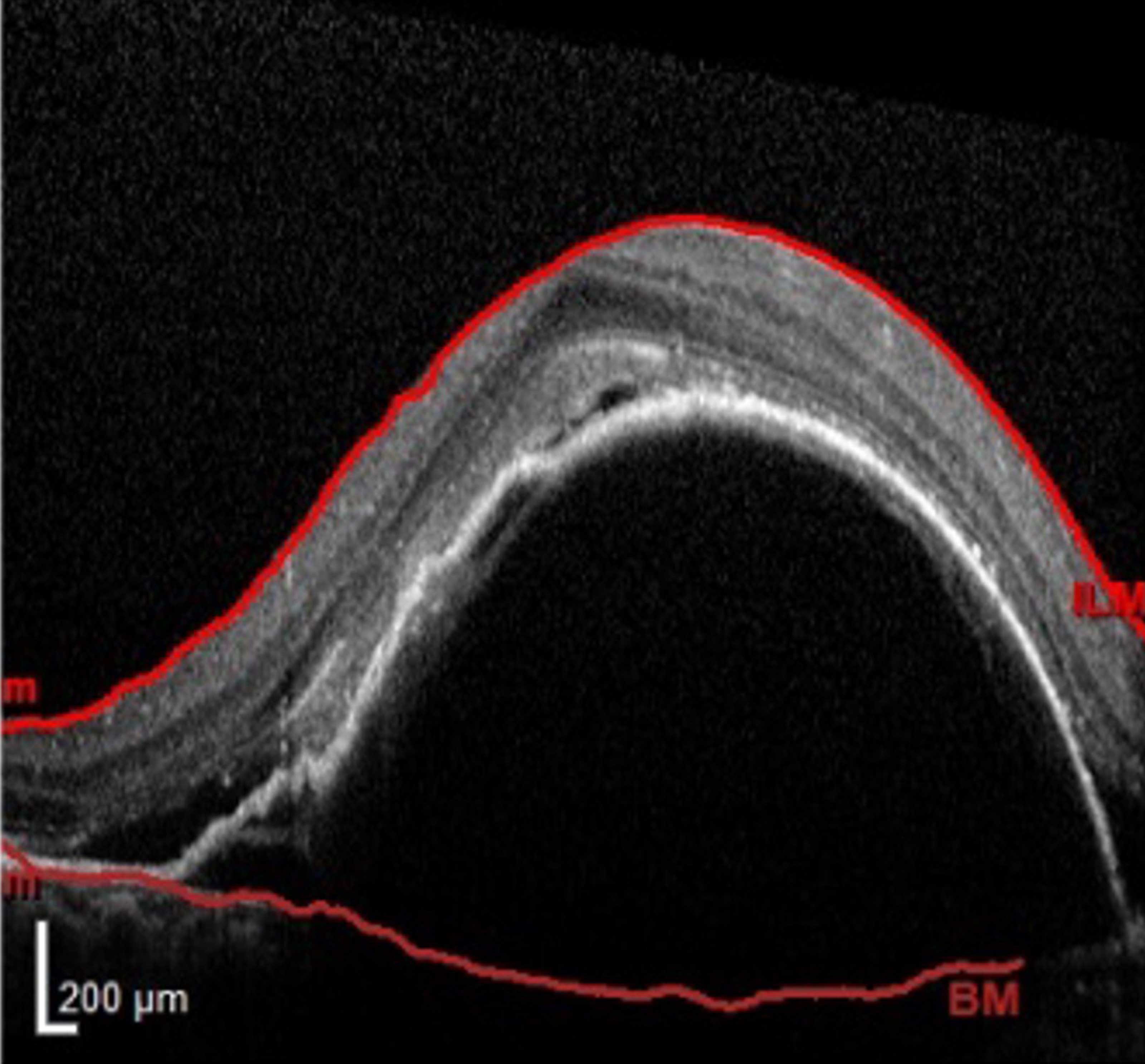
Figure 1: A spectral domain optical coherence tomography image displaying a giant PED with SHRM and SRF before treatment.
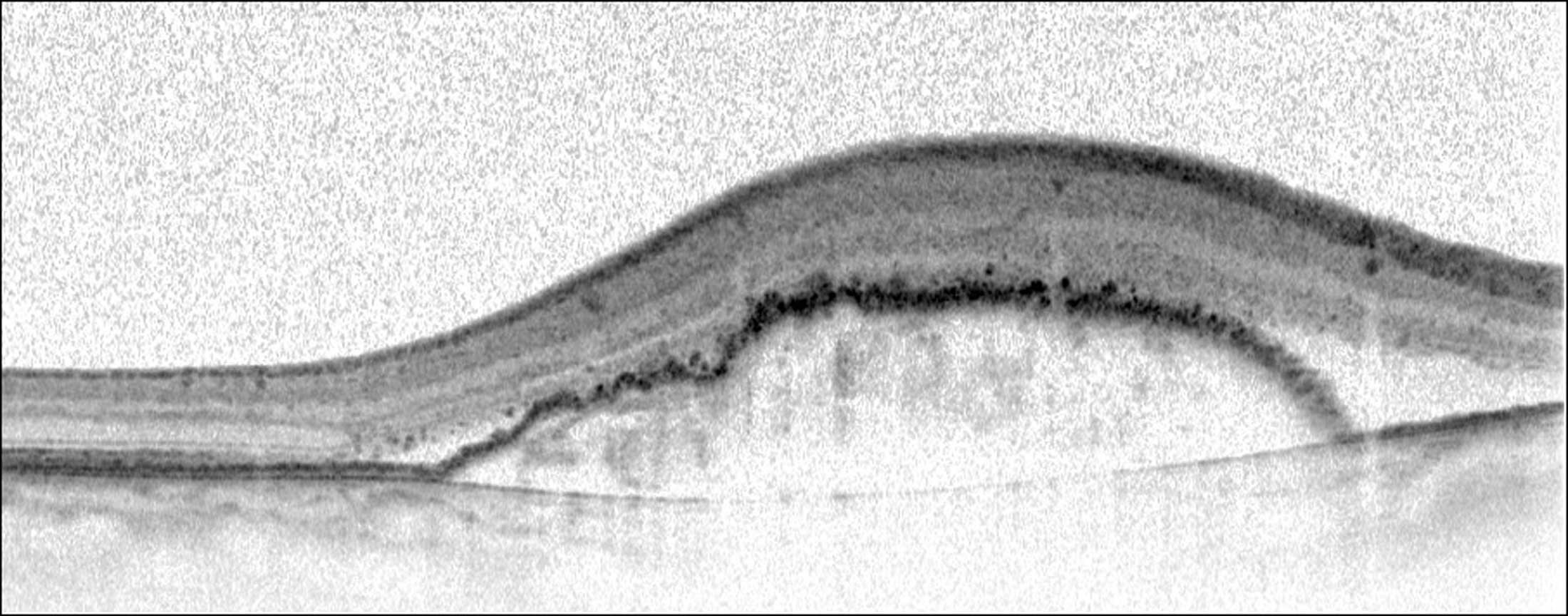
Figure 2: A Spectral-domain optical coherence tomography image before treatment demonstrating a jagged surface on the nasal side of the RPE.
Optical coherence tomography (OCT) findings (right eye) (Figures 1, 2):
- Giant vascularised PED with associated subretinal fluid (SRF)
- Subretinal hyperreflective material (SHRM) on the PED dome
- Jagged RPE surface nasally
- Hyperreflective tissue under the RPE consistent with type 1 macular neovascularisation
- No intraretinal fluid (IRF) or haemorrhage.
PED measurements:
- Height: 1091µm
- Linear diameter: 5112µm
- Central retinal thickness: 703µm
- Central subfoveal volume: 0.55mm³
- Maximum central height (C-Max): 1338µm.
Given the exceptionally high PED height and jagged RPE surface – two major predictors of RPE tear – a half-dose aflibercept injection was chosen to reduce mechanical stress on the RPE. The patient received 1mg (half-dose) intravitreally and returned two weeks later for review.
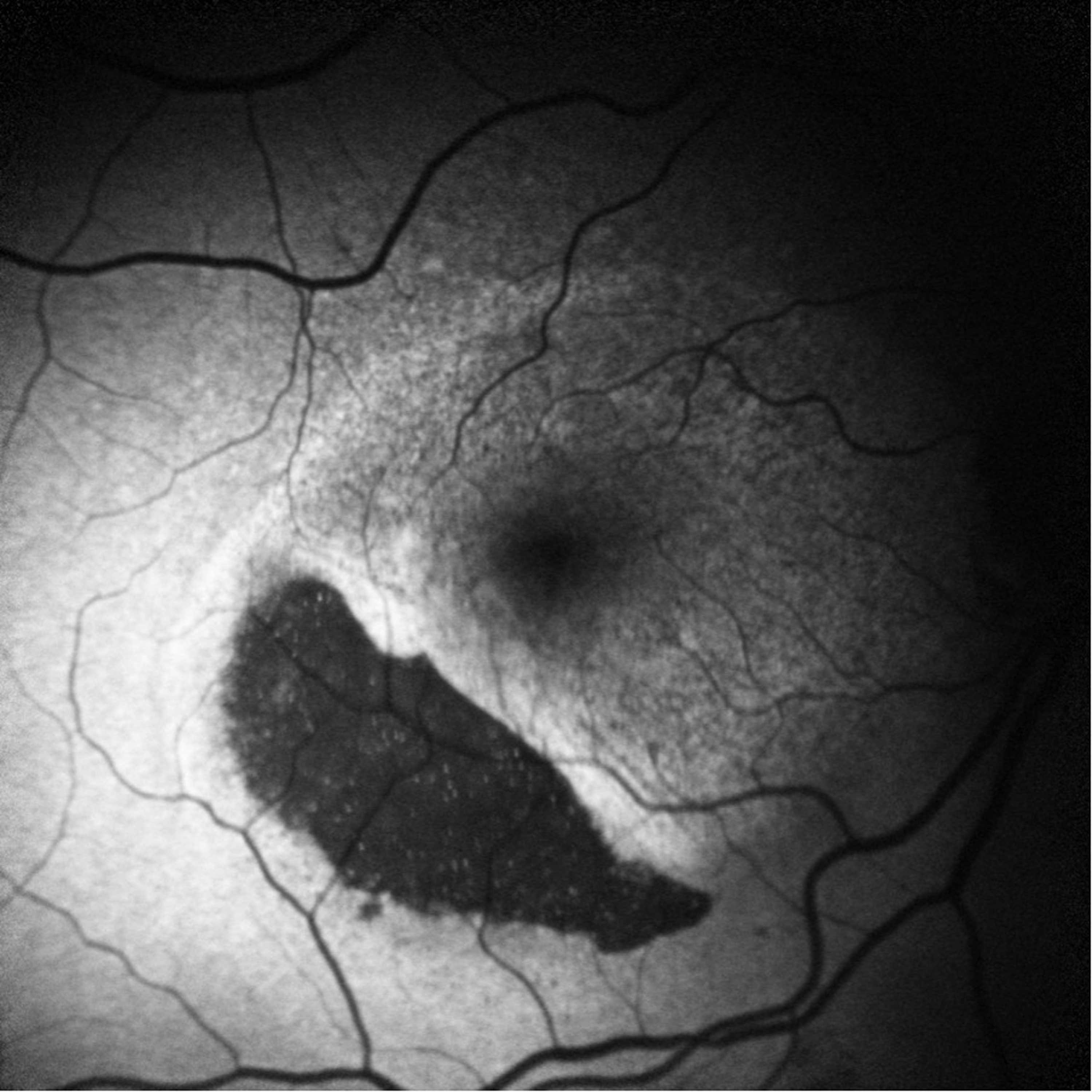
Figure 3: Two weeks after treatment, an autofluorescence image showing a stage 3 RPE tear involving the inferotemporal area.
At the two-week follow-up, BCVA had improved to logMAR 0.48, and OCT demonstrated a marked reduction in PED height and SHRM. However, fundus examination and fundus autofluorescence revealed a grade 3 RPE tear in the inferotemporal region measuring 5112µm, sparing the foveal centre (Figure 3). Although the tear occurred despite dose reduction, its location outside the fovea contributed to maintaining central vision.
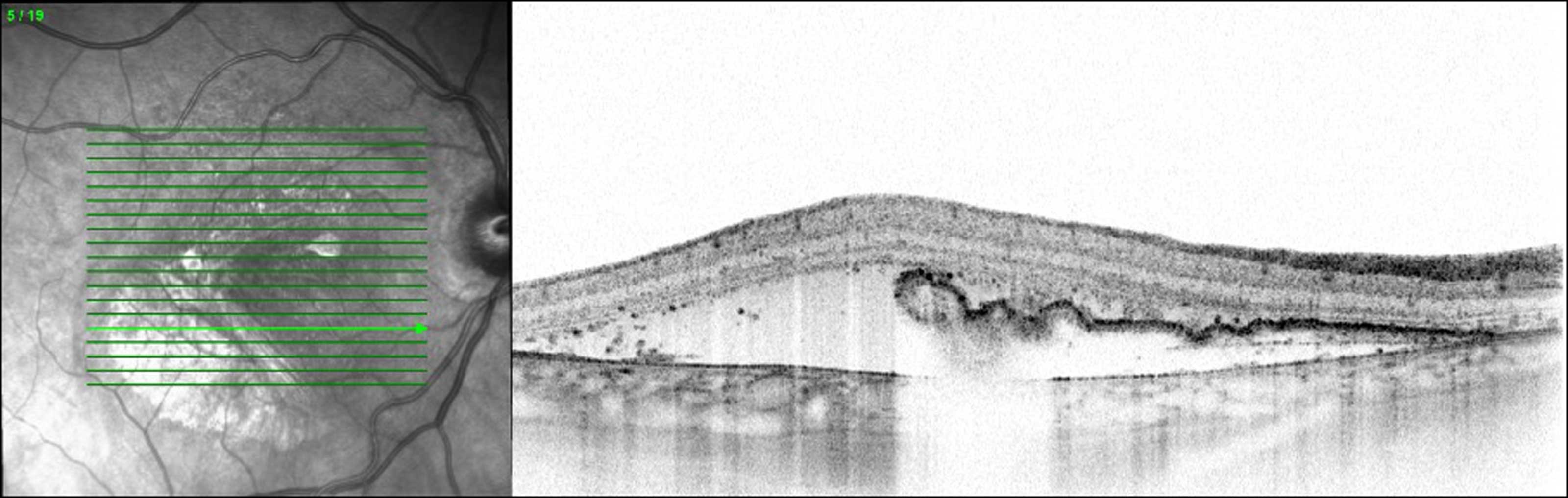
Figure 4: Two weeks after treatment, the optical coherence tomography image revealed a segment devoid of RPE consistent with the RPE tear.
Figure 4 illustrates the OCT appearance after the tear, showing an area of absent RPE corresponding to the defect.
Due to persistent SHRM, a second half-dose aflibercept injection was administered two weeks later. The half-dose regimen was intentionally maintained to avoid further exacerbation of the RPE tear. The patient received four monthly half-dose injections in total, during which the tear remained stable and no further extension was observed.
Following stabilisation, the patient was transitioned to a full-dose aflibercept treat-and-extend regimen. She subsequently received five full-dose injections, with the most recent treatment delivered at an eight-week interval. Nine months after the initial presentation, the BCVA in the right eye remained at logMAR 1.0. During a recent visit, recurrence of SRF and IRF was noted, prompting a reduction in the treatment interval for the tenth aflibercept injection to six weeks.
Discussion
Retinal pigment epithelium tears are a known complication of fibrovascular PEDs associated with nAMD. The spontaneous tear rate in eyes with PED is estimated at around 10%, though this may increase following anti-VEGF therapy. Tears can range from minor, minimally symptomatic disruptions to large, visually devastating defects when the fovea is involved. Real-world studies have reported RPE-tear incidences of 14–17% in vascularised PEDs undergoing anti-VEGF therapy.
Although anti-VEGF agents do not directly induce RPE tears, their mechanism of action contributes to risk: rapid contraction of the macular neovascular membrane following treatment generates acute traction on the overlying RPE, particularly in eyes with large, steep PEDs. In some studies, small tears may have been under-detected, contributing to variability in reported incidence.
The overall incidence of RPE tears in nAMD was comparable across three studies: Chan, et al., (2.2%) [1]; Ronan, et al., (0.8%) [2]; and more recently, Junya N, et al. (1.59%) [3].
Multiple risk factors for RPE tear have been identified, including large PED diameter, increased height, jagged RPE morphology, and sub-RPE hyporeflective spaces. Additional contributing factors include shorter PED duration, vascularised PED, radial hyperreflective lines, and a small ratio of macular neovascular lesion size relative to PED dimensions [4].
The potential benefit of half-dose anti-VEGF therapy for high-risk PEDs has been explored in several reports. Mones, et al. demonstrated the safe flattening of a large PED associated with retinal angiomatous proliferation using biweekly half-dose ranibizumab [5]. Sarraf, et al. reported a higher tear incidence with high-dose ranibizumab, suggesting a dose-response relationship between treatment intensity and risk of tear formation [6]. Nagiel, et al. reported successful resolution of a giant PED measuring 1500µm with a single half-dose aflibercept injection, without RPE tear [7].
These findings suggest that reduced-dose therapy may mitigate contractile forces and lower the risk of tears in some high-risk eyes. Nonetheless, in our case – where PED height exceeded 1000µm and jagged RPE was present – dose reduction did not prevent a tear. However, it may have reduced severity, preventing progression to a grade 4 tear, which would likely have caused profound central visual loss.
This case emphasises that even half-dose anti-VEGF therapy carries substantial risk in extremely high-risk PEDs. Careful risk–benefit assessment and detailed patient counselling are essential before initiating treatment. Adjusting treatment regimens may help reduce the risk of tears [8], but further evidence is required to determine the optimal approach. Continued anti-VEGF therapy is recommended after an RPE tear if exudative activity persists.
References
1. Chan CK, Meyer CH, Gross JG, et al. Retinal pigment epithelial tears after intravitreal bevacizumab injection for neovascular age-related macular degeneration. Retina 2007;27(5):541–51.
2. Ronan SM, Yoganathan P, Chien FY, et al. Retinal pigment epithelial tears after intravitreal injections of bevacizumab (Avastin) injection for neovascular age-related macular degeneration. Retina 2007;27(5):535–40.
3. Nagata J, Shiose S, Ishikawa K, et al. Clinical characteristics of eyes with neovascular age-related macular degeneration and retinal pigment epithelium tears. J Clin Med 2023;12(17):5496.
4. Clemens CR, Eter N. Retinal pigment epithelium tears: risk factors, mechanism and therapeutic monitoring. Ophthalmologica 2016;235(1):1–9.
5. Monés J, Biarnés M, Badal J. Bimonthly half-dose ranibizumab in large pigment epithelial detachment and retinal angiomatous proliferation with high risk of retinal pigment epithelium tear: a case report. Clin Ophthalmol 2013:7:1089–92.
6. Sarraf D, Chan D, Rahimy E, Abraham P. Prospective evaluation of the incidence and risk factors for the development of RPE tears after high- and low-dose ranibizumab therapy. Retina 2013;33(8):1551–7.
7. Nagiel A, Sadda SR, Schwartz SD, Sarraf D. Resolution of a giant pigment epithelial detachment with half-dose aflibercept. Retina Cases Brief Rep 2015;9(4):269–72.
8. Mitchell P, Rodríguez FJ, Joussen AM, et al. Management of retinal pigment epithelium tear during anti-vascular endothelial growth factor therapy. Retina 2020;41(4):671–8.
Declaration of competing interests: None declared.