Geographic atrophy (GA) is an advanced form of age-related macular degeneration (AMD) characterised by progressive, irreversible loss of the retinal pigment epithelium (RPE) and photoreceptors and is estimated to account for approximately 10% of AMD-related blindness [1-4].
The Age-Related Eye Disease Study Research Group found that the median time to developing central GA after any GA diagnosis was two-and-a-half years and that average visual acuity (VA) decreased by 22 letters at year five [5]. Many patients with GA lose the ability to drive within two years, and vision-related quality of life is significantly impacted [3].
Leng, et al., recently published findings on VA and progression risk in patients with dry AMD from an evaluation of the American Academy of Ophthalmology IRIS Registry [6]. Data from 593,277 patients were analysed. Patients with more advanced dry AMD had lower VA at baseline, VA worsening was greater among eyes with better VA at baseline and VA progression was found to be generally faster with each progressive dry AMD stage.
Modelling results showed, on average, that the annual probability of progression from VA ≥20/63 to VA <20/400 was almost tenfold greater among GA with subfoveal involvement study eyes compared with mild dry AMD study eyes (37.7% vs 4%). The annual probability of progression to neovascular AMD was approximately 7-8% among eyes with intermediate to GA with subfoveal involvement at baseline. More than 80% of the study population had bilateral dry AMD, consistent with previous studies confirming high rates of bilateral involvement in AMD.
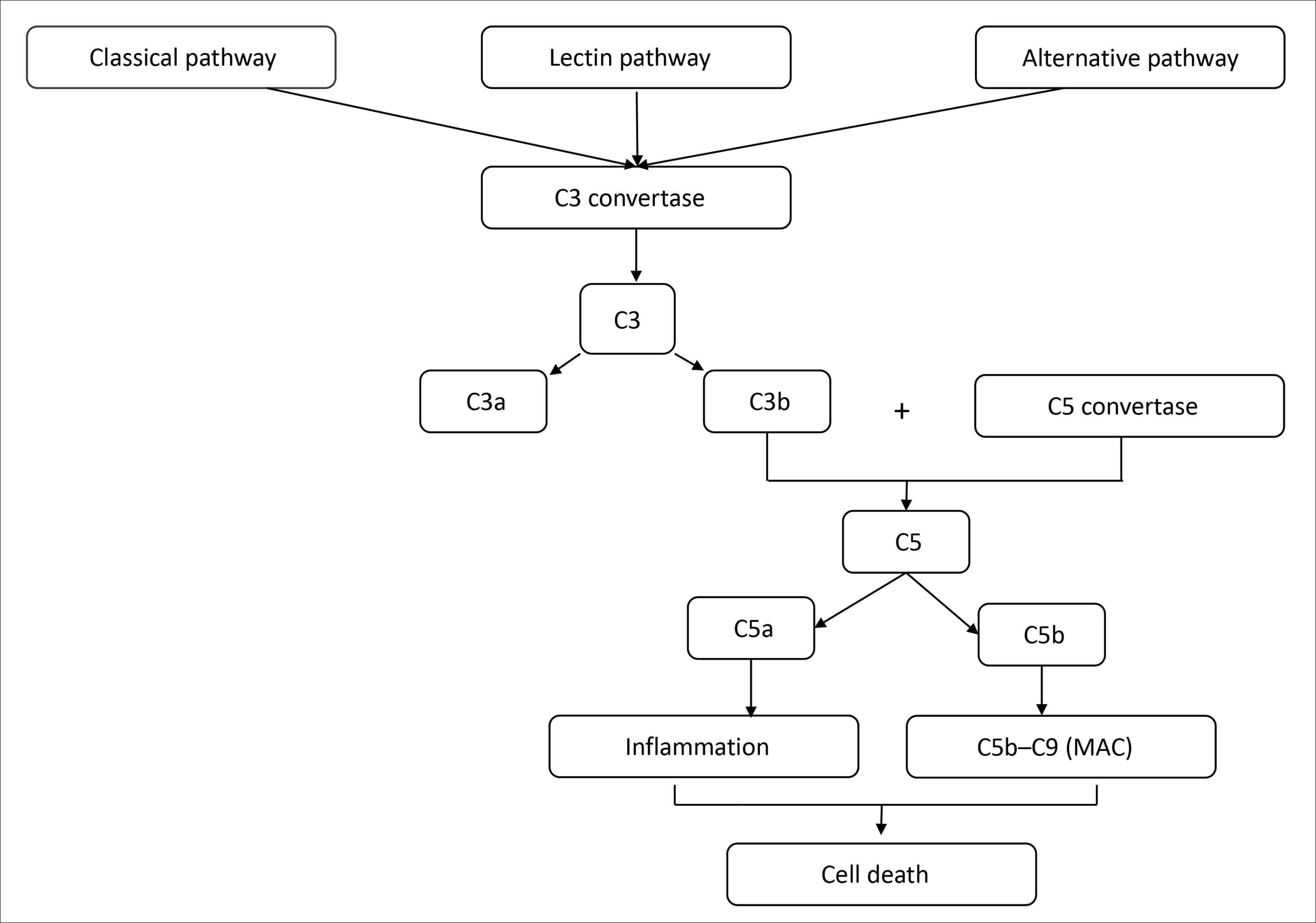
Figure 1: The complement cascade: All three pathways converge at C3 convertase, leading to the cleavage of C3 into C3a and C3b, formation of C5 convertase, cleavage of C5 into C5a and C5b, and eventual formation of the membrane attack complex (MAC) and subsequent cell destruction (Patel 2022). Adapted from: Desai D, et al. Eye 2022;36:294-302; Patel PN, et al. Biomedicines 2022;10:1884.
Dysregulation of the complement cascade and complement inhibition for patients with GA
Dysregulation of the complement cascade is considered a key contributor to the pathophysiology of GA, supported by human genetic findings as well as physiological and preclinical evidence of complement cascade dysfunction in GA (Figure 1) [2,7]. Complement overactivation has been strongly correlated with the development and progression of AMD [1].
US FDA approves first treatment for GA secondary to AMD
Pegcetacoplan injection (Syfovre™, Apellis Pharmaceuticals, Inc.), a complement C3 inhibitor, is the first and only treatment approved in the US by the Food and Drug Administration (FDA) for the treatment of GA secondary to AMD [8]. Pegcetacoplan binds to complement protein C3 and its activation fragment C3b with high affinity thereby regulating the cleavage of C3 and the generation of downstream effectors of complement activation [8]. At 24 months across phase 3 clinical trials (OAKS and DERBY), both monthly and every-other-month pegcetacoplan reduced GA lesion growth with increasing effects over time and showed a well-demonstrated safety profile in ~12,000 injections over two years [9].
Pegcetacoplan is indicated for all patients with GA, with or without subfoveal involvement, with flexible dosing options of once every 25-60 days. Initial priority patients, described in a corporate presentation by Apellis Pharmaceuticals in February 2023, include: significant vision loss in one eye and foveal encroachment in the other eye; nAMD in one eye and GA in the fellow eye; bilateral GA; and nAMD / GA in the same eye. Vial price in the US is $2190, with the majority of patients expected to be covered by Medicare.
Marketing applications for intravitreal pegcetacoplan for the treatment of GA secondary to AMD are under review by the European Medicines Agency (EMA) and regulatory authorities in Canada, Australia, the UK and Switzerland. A decision from the European Commission is expected early 2024 and from other local regulatory authorities in the first half of 2024.
Phase 3 OAKS and DERBY clinical trial results: reduction in mean rate of GA lesion growth observed in both studies
OAKS (n=637) and DERBY (n=621) were phase 3, multicentre, randomised, double-masked, sham-controlled studies comparing the efficacy and safety of pegcetacoplan injection with sham injections across a broad and heterogenous population of patients with GA secondary to AMD [8,9]. The studies evaluated the efficacy of monthly and every-other-month pegcetacoplan assessed by change in the total area of GA lesions from baseline as measured by fundus autofluorescence.
REDUCTION IN LESION GROWTH RATE WITH PEGCETACOPLAN (SyfovreTM) VS SHAM POOLED 1*†
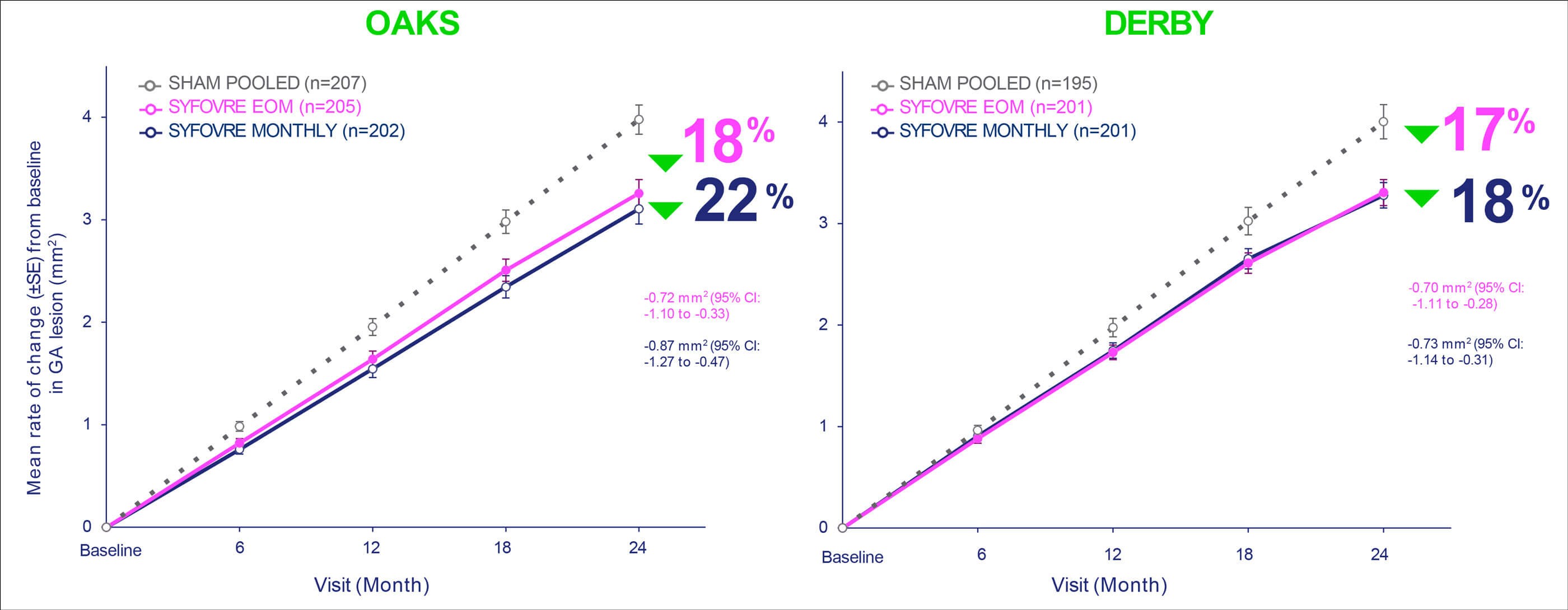
Figure 2: Treatment with every-other-month and monthly pegcetacoplan injection (SyfovreTM) resulted in a reduction of GA lesion growth across both studies: *Based on a mixed effects model for repeated measures assuming a piecewise linear trend in time with knots at Month 6, Month 12, and Month 18.1 Slope for baseline to Month 24 is an average of slope of baseline to Month 6, Month 6 to Month 12, Month 12 to Month 18, and Month 18 to Month 24.1 †SHAM EOM and SHAM Monthly were pooled for analysis.2 confidence interval (CI); every other month (EOM); standard error (SE). 1. SYFOVRE (pegcetacoplan injection) [package insert]. Waltham, MA: Apellis Pharmaceuticals, Inc; 2023. 2. Data on file, Apellis Pharmaceuticals, Inc. Adapted from: SYFOVRE™ (pegcetacoplan injection) FDA Approval: Conference Call, February 17, 2023. Apellis Pharmaceuticals.
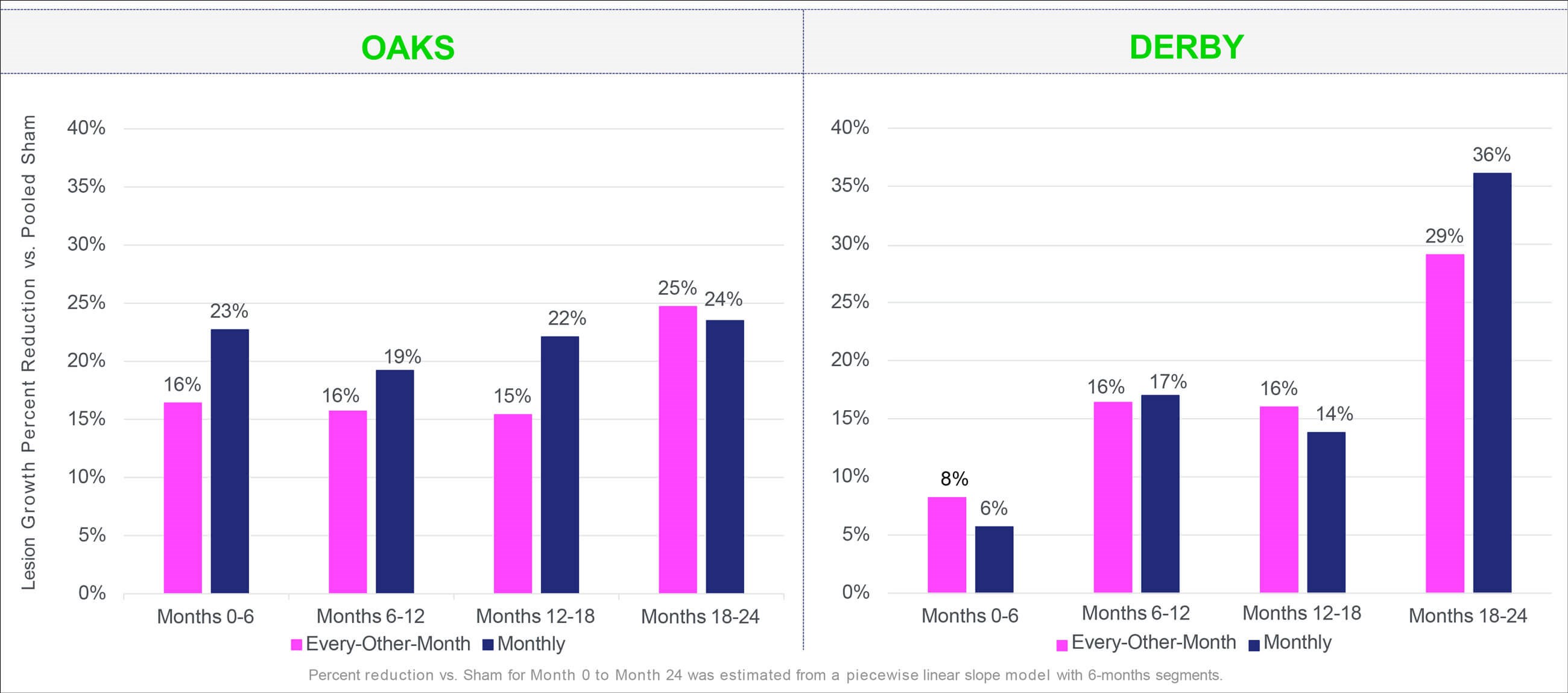
Figure 3: Pegcetacoplan injection (SyfovreTM) demonstrated increased effects over 24 months: Percent reduction vs. sham for Month 0 to Month 24 was estimated from a piecewise linear slope model with six-month segments. Adapted from: SYFOVRETM (pegcetacoplan injection) FDA Approval: Conference Call, February 17, 2023. Apellis Pharmaceuticals.
Treatment with pegcetacoplan monthly or every-other-month resulted in a reduction of GA lesion growth versus sham at month 24 across both studies (OAKS: 22% monthly, p<0.0001; 18% bimonthly, p=0.0003; DERBY: 18% monthly, p=0.0006; 17% bimonthly, p=0.0010) (Figure 2 and Figure 3). Reductions in GA lesion growth were consistent irrespective of lesion location. Although no statistically significant differences were observed in key functional endpoints, including best-corrected visual acuity (BCVA) and microperimetry (OAKS only), a signal of functional benefit was observed in the post-hoc microperimetry analyses of the junctional zone.
Pegcetacoplan was well tolerated over 24 months, with most ocular study eye adverse events being mild to moderate. The most common adverse events (incidence ≥5%) are ocular discomfort, nAMD, vitreous floaters and conjunctival haemorrhage. By month 24, rates of new onset neovascular AMD in the study eye were 12.2% for pegcetacoplan monthly, 6.7% for pegcetacoplan bimonthly and 3.1% in the control group (OAKS and DERBY combined). The majority (85.7%) of exudative AMD lesions were classified as occult. The GALE extension study (NCT04770545) will report the long-term impact of treatment with pegcetacoplan in patients with GA.
Reductions in both RPE and photoreceptor degeneration were observed after intravitreal pegcetacoplan therapy in the OAKS and DERBY phase 3 trials
Post-hoc analyses from the OAKS and DERBY studies, reported at the Association for Research in Vision and Ophthalmology (ARVO) annual meeting in April 2023, showed visual function and quality-of-life benefits in pegcetacoplan-treated patients with extrafoveal lesions. Pegcetacoplan also slowed photoreceptor (PR) and RPE cell loss compared with sham, with both PR and RPE degeneration significantly and consistently reduced after 24 months on intravitreal pegcetacoplan therapy in both trials (p-values nominal) [10]:
- PR loss reductions vs sham: every other month: 46% (OAKS; p<0.0001) and 46% (DERBY; p<0.0001); monthly: 53% (OAKS; p<0.0001) and 47% (DERBY; p<0.0001); and
- RPE loss reductions vs sham: every-other-month: 20% (OAKS; p=0.0002) and 21% (DERBY; p=0.0005); monthly: 22% (OAKS; p=0.0002) and 27% (DERBY; p<0.0001).
Efficacy of avacincaptad pegol in GA
Avacincaptad pegol (ACP) (Zimura®, IVERIC Bio, Inc.) is an investigational pegylated RNA aptamer designed to be a specific inhibitor of complement 5 [11].
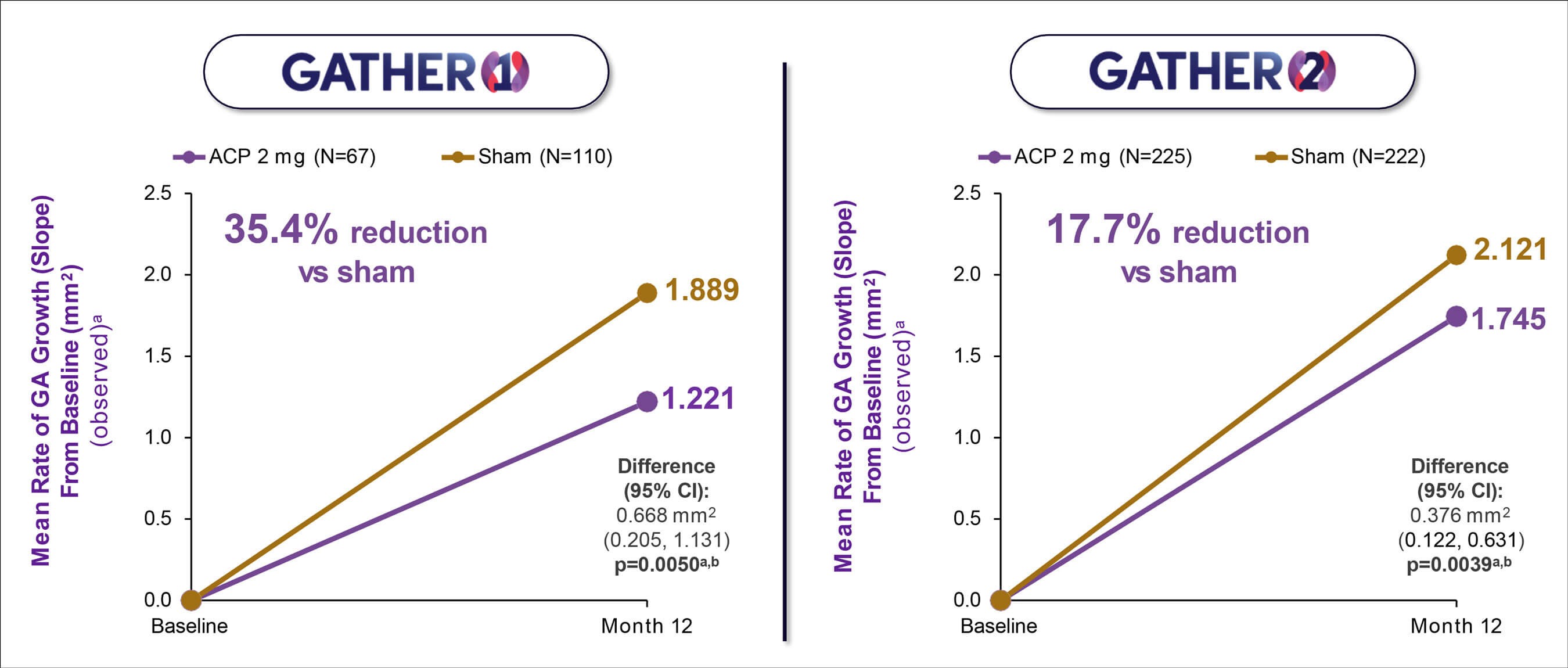
Figure 4: GATHER 1 and GATHER 2: Mean rate of observed GA growth (slope analysis) demonstrated consistent efficacy results between the two studies. Note: The primary analysis for GATHER1 (mean change in square root transformed GA area from baseline to month 12 [mm]) is consistent with the slope analysis utilising observed data. The estimates for the GATHER1 ACP 2 mg group vs sham are from the MMRM model, drawing on all available data, including data from groups with different randomisation ratios in Part 1 and Part 2 of the trial, and should not be interpreted as directly observed data. a Non-square root transformation; b Descriptive p-value. Avacincaptad pegol (ACP); confidence interval (CI); geographic atrophy (GA). Data on file. IVERIC Bio. Khanani KM, et al. Efficacy of avacincaptad pegol in geographic atrophy: GATHER1 and GATHER2 Results. Presentation at The Retina Society 55th. Annual Scientific Meeting, Pasadena, CA, November 2-5, 2022.
GATHER1 phase 2/3 and GATHER2 phase 3 are multicentre, prospective, randomised, double-masked, sham-controlled trials designed to evaluate the efficacy and safety of monthly intravitreal avacincaptad pegol vs sham in patients with GA (Figure 4) [11]. Enrolled participants had to have disease in part within 1500 microns of but not involving the centre point. The primary efficacy endpoints in both studies were based on GA area measured by fundus autofluorescence at three time points: baseline, month six, and month 12.
In GATHER1, for the primary endpoint assessment (square root transformation) at 12 months, there was a 27.4% (p=0.0072) reduction in the mean rate of growth (slope analysis) estimated based on GA area between the ACP 2mg group and the corresponding sham control group over 12 months. Using observed data, there was a 35% (p=0.0050) reduction in the mean rate of growth (slope analysis) estimated based on GA area between the two groups over 12 months. Treatment with ACP 2mg was associated with continued reductions in the progression of GA growth compared with sham through 18 months [12].
In GATHER2, there was 14.3% (p=0.0064) reduction in the mean rate of growth (slope analysis) in GA area between the two groups at 12 months with the primary analysis (square root transformation) and a 17.7% (p=0.0039) reduction in the mean rate of growth (slope analysis) in GA area between the two groups at 12 months using the observed GA area [13].
Through 12 months, the incidence of choroidal neovascularisation (CNV) in the study eye was 9% in the ACP 2mg group and 2.7% in the corresponding sham control group in GATHER1. Over the 18-month study period, macular neovascularisation conversion rates were 11.9% with ACP 2mg and 2.7% in the corresponding sham control group [12]. In GATHER 2, the incidence of CNV in the study eye was 6.7% in the ACP 2mg group and 4.1% in the sham control group in year one.
An exploratory time-to-event analysis from the GATHER trials signalled a 44% reduction (Hazard Ratio 0.56 with 95% CI, 0.15-2.06) and a 59% reduction (Hazard Ratio 0.41 with 95% CI, 0.17-1.00) respectively in the rate of vision loss (≥15 letters from baseline measured at two consecutive visits up to month 12) with ACP 2mg compared with sham over the first 12 months of treatment.
In February 2023, IVERIC bio, Inc. announced that the FDA had accepted the company’s Non-Disclosure Agreement (NDA) for approval of ACP for the treatment of GA secondary to AMD, with a Prescription Drug User Fee Act target action date in August 2023.
Investigational treatments for GA in phase 2 clinical trials
Several other companies are actively developing complement modulation-based product candidates for the treatment of GA (Table 1).
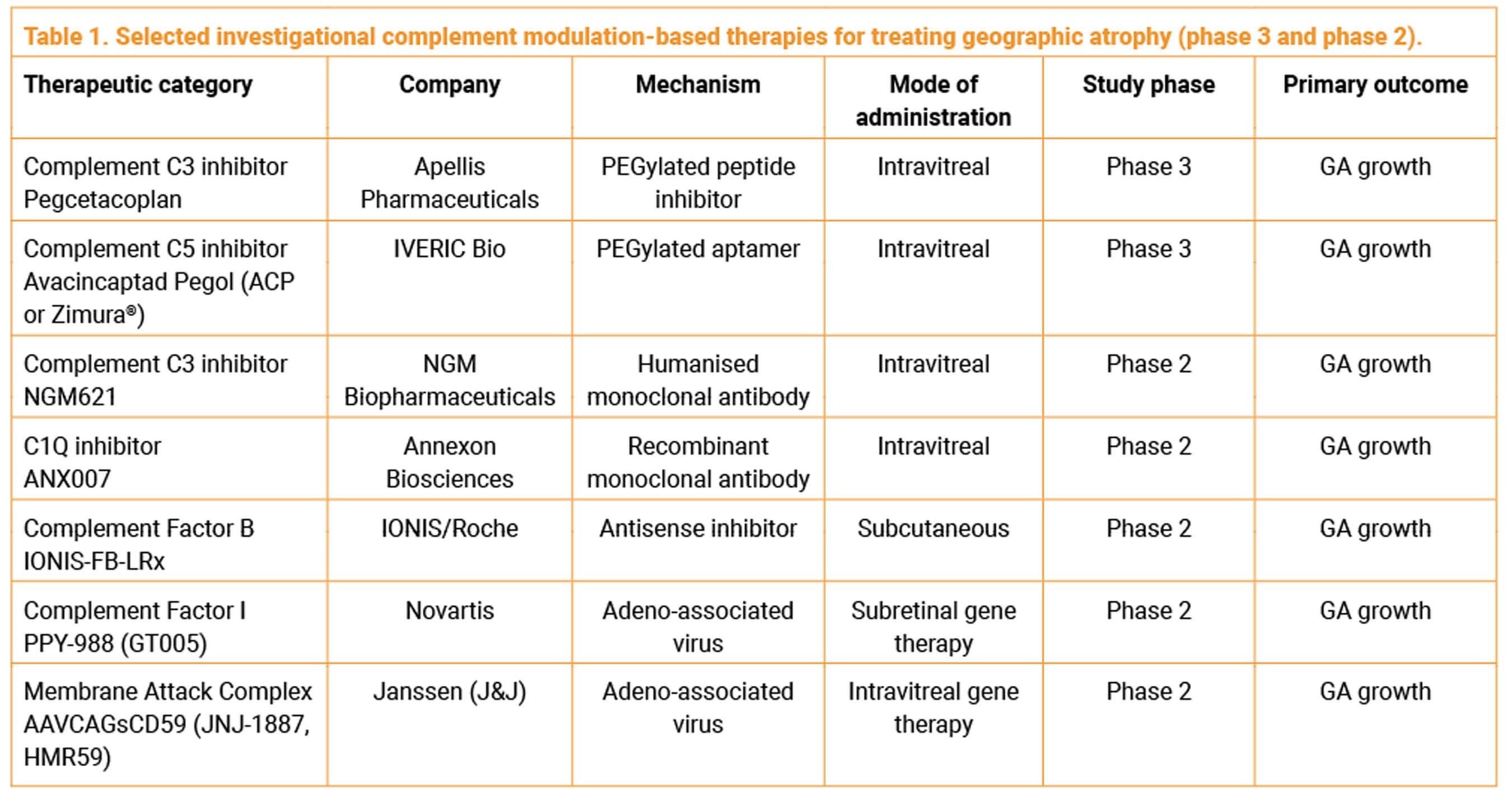
NGM621 (NGM Biopharmaceuticals, Inc.) is a monoclonal antibody that inhibits complement factor C3. The phase 2 CATALINA trial, completed late last year, did not meet its primary endpoint of a statistically significant rate of change using slope analysis in GA lesion area over 52 weeks for NGM621 versus sham. Over 52 weeks, intravitreal NGM621 given every four weeks (Q4W) (n=108) and every eight weeks (n=104) demonstrated a reduction in the rate of change in GA lesion area (slope) of 6.3% and 6.5%, respectively, compared to sham (n=106), which did not reach statistical significance in either arm. Additional post-hoc analysis that adjusted for large lesion variability showed potentially encouraging findings warranting further evaluation. In a pre-specified secondary mixed effects model for repeated measures (MMRM) analysis, NGM621 Q4W showed a treatment effect at 24 weeks (nominal p<0.05) that diminished by 52 weeks (19.8% reduction in GA lesion area at week 24, 7.4% at over 52 weeks). NGM621 showed no evidence of increased CNV conversion vs sham and was well-tolerated with no treatment-related serious adverse events. The CNV conversion rate in the fellow eye was 4.2% over 56 weeks.
Intravitreal ANX007 (Annexon Biosciences, Inc,) is a novel C1q inhibitor being evaluated in a global phase 2 clinical trial (ARCHCER) in patients with GA. ANX007 is an antigen-binding fragment (Fab) designed to block C1q locally in the eye, to provide more complete protection against excess classical complement activity. The company anticipates reporting initial clinical trial data in mid-2023, with additional data to be presented after the conclusion of the six-month off-treatment period by the end of 2023. In a phase 1b trial, ANX007 was well-tolerated and demonstrated full target engagement and inhibition of C1q in the eye for at least four weeks. Intravitreal ANX007 protected photoreceptor cells and retinal function in published preclinical studies. An extended-release formulation is also being assessed.
ASO factor B (IONIS-FB-LRx, RG6299, Ionis Pharmaceuticals, Inc./Roche) is an antisense oligonucleotide that inhibits complement factor B gene expression by binding with factor B mRNA. ASO factor B, administered subcutaneously every four weeks, is being evaluated in a phase 2 clinical trial (GOLDEN STUDY) for the treatment of patients with GA secondary to AMD. The primary outcome is absolute change from baseline in the GA area at week 49, as assessed by retinal imaging.
JNJ-81201887 (JNJ-1887, Jenssen Pharmaceuticals), formerly referred to as AAVCAGsCD59 (HMR59), is an investigational one-time gene therapy for the treatment of GA. JNJ-1887 is designed to increase the expression of a soluble form of CD59 (sCD59) intended to protect retinal cells, thus slowing and preventing disease progression. The phase 2b PARASOL clinical trial is actively enrolling patients to study JNJ-1887 for the treatment of adults aged 60 or older with advanced dry AMD with GA. JNJ-1887 has been granted Fast Track designation by the US FDA and Advanced Therapy Medicinal Product (ATMP) designation by the EMA.
PPY-988 (GT005) (Novartis), currently in phase 2 clinical development, is designed as an AAV2-based, one-time investigational gene therapy for GA secondary to AMD that is delivered by subretinal injection and designed to increase production of the complement factor I (CFI) protein. It is believed that increasing CFI production could reduce inflammation and potentially help preserve vision.
Research from ARVO 2023 explores vision loss in GA and patient acceptability of potential intravitreal treatment
- A retrospective study of VA changes and conversion to nAMD over three years in 18,712 eyes with GA found that patients lost two lines of vison at year three and 25% were diagnosed with nAMD during follow-up [14]. Study authors suggested that those GA eyes with modest VA impairment at baseline may be at the greatest risk of further vision loss, due largely to progression to subfoveal GA.
- Retrospective analysis of patients with bilateral GA enrolled in the lampalizumab trials found that a more rapid rate of BCVA loss was seen with a higher GA lesion growth rate as compared with a lower growth rate [15].
- A qualitative study of patient acceptability of intravitreal injection therapy for GA, conducted in a retina clinic in London and involving 30 participants with GA, reported that around three in every five participants (60%) would accept intravitreal treatment that could slow but not halt GA progression, with 40% undecided or negative [16]. Perceived limited magnitude of treatment efficacy, increased risk of nAMD, and burden of regular clinic visits were the main factors limiting acceptability.
Debate: complement inhibition is a ‘game changer’ in the management of patients with GA
In a debate on potentially ‘game-changing’ complement inhibitors for GA at the 14th Annual Congress on Controversies in Ophthalmology in Lisbon in March 2023, Peter K Kaiser from USA noted that complement inhibition has been shown to slow GA progression and to slow photoreceptor loss in junctional zone around GA, verified by microperimetry. The effect on structure and function is expected to increase over time and he argued that the side-effect profile in GA patients treated with complement inhibitors is manageable. Ursula Schmidt-Erfurth from Austria emphasised that preservation of RPE and PR integrity is the true ‘game’.
References
1. Holz FG, Strauss EC, Schmitz-Valckenberg S, van Lookeren Campagne M. Geographic atrophy: clinical features and potential therapeutic approaches. Ophthalmol 2014;121(5):1079-91.
2. Boyer DS, Schmidt-Erfurth U, van Lookeren Campagne M, Henry EC, Brittain C. The pathophysiology of geographic atrophy secondary to age-related macular degeneration and the complement pathway as a therapeutic target. Retina 2017;37(5):819-35.
3. Chakravarthy U, Bailey CC, Johnston RL, et al. Characterizing disease burden and progression of geographic atrophy secondary to age-related macular degeneration. Ophthalmol 2018;125(6):842-9.
4. Sunness JS, Rubin GS, Applegate CA, et al. Visual function abnormalities and prognosis in eyes with age-related geographic atrophy of the macula and good visual acuity. Ophthalmol 1997;104(10):1677-91.
5. Lindblad AS, Lloyd PC, Clemons TE, et al. Age-Related Eye Disease Study Research Group. Change in area of geographic atrophy in the Age-Related Eye Disease Study: AREDS report number 26. Arch Ophthalmol 2009;127(9):1168-74.
6. Leng T, Schwartz J, Nimke D, et al. Dry age-related macular degeneration: distribution of visual acuity and progression risk in a large registry. Ophthalmol Ther 2023;12(1):325-40.
7. Desai D, Dugel PU. Complement cascade inhibition in geographic atrophy: a review. Eye (Lond) 2022;36(2):294-302.
8. SYFOVRE™ Prescribing Information. Apellis Pharmaceuticals, Inc.
9. Lad EM. 2-year outcomes of the pegcetacoplan phase 3 Derby and Oaks clinical trials for GA: baseline characteristics and efficacy. Presentation at the Angiogenesis, Exudation, and Degeneration 2023 Virtual Meeting, 10-11 February 2023.
10. Schmidt-Erfurth U, Julia Mai J, Reiter GS, et al. Therapeutic effect of pegcetacoplan on retinal pigment epithelium (RPE) and photoreceptor (PR) integrity in geographic atrophy (GA) in the phase 3 OAKS and DERBY trials. ARVO 2023 Annual Meeting, New Orleans, LI, USA; 23-27 April 2023.
11. Khanani AM. The efficacy of avacincaptad pegol in geographic atrophy: GATHER1 and GATHER2 Results. Presentation at the Retina Society 55th Annual Scientific Meeting, Pasadena, CA, USA; November 2-5, 2022
12. Patel SS, Lally DR, Hsu J, et al. Avacincaptad pegol for geographic atrophy secondary to age-related macular degeneration: 18-month findings from the GATHER1 trial. Eye (Lond) 2023 Mar 24. Epub ahead of print. .
13.Lally D. C5 inhibition with avacincaptad pegol for the treatment of geographic atrophy: efficacy results from the phase 3 GATHER2 trial. Presentation at the Angiogenesis, Exudation, and Degeneration 2023 Virtual Meeting, February 10-11, 2023.
14.Ciulla TA, Boucher N, Aggarwal N, et al. Geographic atrophy is associated with meaningful disease burden: visual acuity changes and conversion to neovascular AMD over 3 years in 18,712 patient eyes. ARVO 2023 Annual Meeting, New Orleans, LI, USA; April 23-27, 2023.
15.Steffen V, Anegondi N, Daniela Ferrara D, et al. Visual function loss in geographic atrophy: learnings from lampalizumab trial data. ARVO 2023 Annual Meeting, New Orleans, LI, USA; April 23-27, 2023.
16.Dinah C, Enoch J, Ghulakhzsian A, et al. Patient acceptability of intravitreal therapies for Geographic atrophy: a mixed-methods study. ARVO 2023 Annual Meeting, New Orleans, LI, USA; April 23-27 2023.
COMMENTS ARE WELCOME






