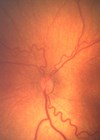
A male child is born at 31 weeks and requires 100% oxygen supplementation with intensive care. What would be the ophthalmic management of this child?
In the first instance, it will be useful to know the screening criteria and protocol. An evidence-based guideline for the screening and treatment of retinopathy of prematurity (ROP) was developed by a multidisciplinary guideline development group (GDG) of the Royal College of Paediatrics & Child Health (RCPCH) in collaboration with the Royal College of Ophthalmologists (RCOphth), British Association of Perinatal Medicine (BAPM) and the premature baby charity BLISS, which is as follows:
Screening criteria: All babies less than 32 weeks gestational age (up to 31 weeks and six days) or less than 1501g birth-weight should be screened for ROP.
Screening protocol: Babies born before 27 weeks gestational age (i.e. up to 26 weeks and six days) – the first ROP screening examination should be undertaken at 30 to 31 weeks postmenstrual age.
Babies born between 27 and 32 weeks gestational age (i.e. up to 31 weeks and six days) – the first ROP screening examination should be undertaken between four and five weeks (i.e. 28-35 days) postnatal age.
Babies >32 weeks gestational age but with birthweight <1501 grams – the first ROP screening examination should be undertaken between four and five weeks (i.e. 28-35 days) postnatal age.
Frequency of screening: Minimum frequencies of screening should be weekly when:
- the vessels end in zone I or posterior zone II
- there is any plus or pre-plus disease
- there is any stage 3 disease in any zone
Minimum frequencies of screening should be every two weeks in all other circumstances until the criteria for termination have been reached.
All babies <32 weeks gestational age or birthweight <1501g should have their first ROP screening examination prior to discharge.
Where a decision is made not to screen a baby, the reasons for doing so should be clearly stated in the baby’s medical record and the examination should be rescheduled within one week of the intended examination.
Termination of ROP screening
Screening can be stopped when a baby is no longer at risk of sight-threatening ROP.
In babies who never develop any ROP, the risk of sight-threatening ROP developing is minimal once the retinal vessels have entered zone III. That vessels are in zone III can be difficult to determine, but it is unlikely to occur before 37 weeks postmenstrual age and a decision to stop screening before this must be carefully evaluated.
- In babies without ROP, there is minimal risk of developing sight-threatening ROP when vascularisation has extended into zone III and eye examinations may be stopped when this happens, usually after 36 completed weeks’ postmenstrual age.
- In babies developing ROP which does not meet the criteria for treatment, screening can be safely stopped when there are clear signs that the active progression of ROP has halted and regression has commenced.
- In the presence of ROP, screening for progressive active disease may be discontinued when any of the following characteristics of regression are seen on at least two successive examinations:
• lack of increase in severity
• partial resolution progressing towards complete resolution
• change in colour in the ridge from salmon pink to white
• transgression of vessels through the demarcation line
• commencement of the process of replacement of active ROP lesions by scar tissue.
Post treatment review
Postoperative review is important to monitor disease regression and to determine if re-treatment is necessary.
- The first examination post treatment should take place five to seven days after treatment and should be continued at least weekly for signs of decreasing activity and regression.
- Re-treatment should be performed usually 10–14 days after initial treatment when there has been a failure of the ROP to regress.
Follow-up after screening or treatment
- After the acute phase, eyes that have reached stage 3 or have been treated should be monitored at a frequency dictated by the clinical condition to determine the risk of sequelae.
Mr B Manoj MS; FRCS, Consultant Ophthalmic Surgeon, Wolverhampton Eye Infirmiry.
TAKE HOME MESSAGE
The most important aim is to visualise the peripheral retina following the protocol, and the assessment of the number of clock hours of disease will decide whether or not the baby requires laser.
Screening neonates for retinopathy of prematurity involves both competent use of the indirect ophthalmoscope (hatty) delicate handing of small eyes and manoeuvring around equipment.
Most units have an ROP diary where the paediatricians diarise the names of the babies to set the date of initial screening. Babies’ eyes should have been dilated at a designated time prior to the arrival of the screening ophthalmologist, with drops that are written up by the paediatricians. The ophthalmologist’s responsibility is to indicate the on-going screening interval which is dependent upon their findings at retinal examination and can easily be done by sticking a PID sticker on the next date for screening. Babies react differently to screening and the nurses should make the screener aware of any babies who do not cope well with being handled. Babies may be too sick to be screened at the desired time and subsequent screening should be rearranged as soon as possible after the desired date. Transfers of babies between units is common, and good communication will ensure that screening occurs at appropriate time intervals.
Infection control
The babies have an immature immune system and should be considered immune-compromised, meaning cross infections can occur easily if due care is not taken with hand washing and fomites such as the 20D and 28D lenses and the hatty. Babies with hospital acquired and antibiotic resistant infections should be seen last to decrease transference of infection.
Phenylephrine 2.5% and Cyclopentolate 0.5% are the recommended eye drop regime in the UK. When pupils are poorly dilated up to three doses at five minute intervals is permissible but higher concentrations have recorded side-effects. Dilation of pupils in small babies may need up to an hour and partially dilated pupils (more often seen in those with severe ROP) will compromise the examination of the peripheral retina.
The screening
Babies in open cots are easily accessed, whilst incubators make positioning the baby more difficult.
The placement of the speculum and the bright light of the indirect ophthalmoscope can cause a strong Bells reflex making the baby more difficult to screen than with gentle handling of the eyelids. Holding the small eye-lids apart takes a good amount of practice but there is evidence that it is less distressing for the baby. The most important aim is to visualise the peripheral retina and this may require both a speculum and indenter in a difficult baby where the assessment of the number of clock hours of disease will decide whether or not the baby requires laser.
To optimally visualise the peripheral retina the screener should stand on the opposite side of the baby for the temporal retina and the same side for the nasal retina. In order to move the light peripherally the screener should start by looking straight through the pupil focusing on the posterior retina and slowly rotate the baby’s head and lens away from themselves in order to change the angle of the eye so that the peripheral retina comes into view. The dolls head movements, stimulated by rocking the head from side to side can be used to rotate the eyes into a better position. Using the RetCAM, with a speculum, is less stressful than the indirect ophthalmoscope and gives the best level of documentation of ROP, especially in babies who are on C-PAP where the ventilation equipment impedes visualisation of the temporal retina with the hatty.
Documentation should include personal identity label, gestation at birth, birth weight, date of screening and age at screening. Most units use diagrams to record stages and areas (zones) of disease, the extent of which is represented in clock hours. The presence of pre-plus or plus disease is an indicator of severity and will highlight those babies which require closer observation or treatment.
The small percentage who meet treatment criteria should be treated within 48 hours in established units that are familiar with the procedure and have a designated laser area. The person performing the treatment should be experienced in treating ROP with the trans-pupillary diode laser therapy recommended as the first line treatment. The narrow 48 hour treatment window compounds difficulties in availability of beds, staff and treating ophthalmologists, emphasising the need for comprehensive care pathways to be in place in every neonatal unit.
Miss F Dean FRCOphth, Consultant Ophthalmologist, University Hospital Coventry & Warwick.
COMMENTS ARE WELCOME