Nurse-led intravitreal injections have become of great importance for busy eye units. In this article, the authors present safety data from five years of a nurse-led service with the use of the Precivia® intravitreal injection assist device.
Intravitreal injections of anti-vascular endothelial growth factors (anti-VEGF) are the commonest outpatient procedures completed in the UK, with an estimated 380,000 injections performed annually [1]. This number of injections continues to rise exponentially, owing, in part, to the approval of ranibizumab and aflibercept by National Institute of Health & Care Excellence (NICE).
Solely relying on clinicians for this service cannot meet this increasing demand without compromising patient safety, so the Royal College of Ophthalmologists recommended training allied health professionals, including technicians and nurses, to administer these injections [2].
Following the success of this execution across several hospitals in the UK [3-5], a similar nurse-led intravitreal injection service was introduced at the Great Western Hospital in 2015 incorporating the use of an injection assist device called Precivia® (FCI Ophthalmics, USA). The device is a transparent polycarbonate mould that fits the limbus with a central window enabling patient fixation. A 28° angled guided port facilitates entry with a 30-gauge needle at a fixed depth of 5.6mm to deliver anti-VEGF therapy in a consistent and repeatable manner. We aim to retrospectively review the safety and efficacy of the Precivia® injection device for intravitreal anti-VEGF delivery by suitably trained ophthalmic nurses.
Methods
Recruitment
Patients were retrospectively recruited from the injection clinic at the Great Western Hospital in Swindon in the period from May 2015 to May 2020. Inclusion criteria consisted of all patients who received intravitreal anti-VEGF injections. Patients who were excluded (where Precivia® was not used) were:
- Patients with previous history of trabeculectomy
- Recent cataract surgery (within four weeks)
- Previous history of keratoplasty.
Intravitreal injection procedure 5% povidone-iodine solution is first used for periocular antisepsis, following which the Precivia® device is carefully positioned onto the limbus, ensuring its flanges avoid contact with the eyelashes. Gentle pressure is applied to enhance an anaesthetic effect and the device is rotated to displace the conjunctiva. A 30-gauge needle is guided through the injection port at a fixed angle and depth. Once injected, the needle is withdrawn and the device is rotated to replace and seal the displaced conjunctiva over the injection site.
Data collection
Data including patient demographics, ocular co-morbidities, indication for treatment, injections and complications were collected from Medisoft electronic patient record (EPR).
Data analysis
All analyses were carried out using GraphPad Prism® 6 (GraphPad Software, California, USA) and SPSS ® Statistics 23 (IBM, New York, USA). Shapiro-Wilk test was performed to assess normality of data. Continuous variables are expressed as mean ± SD for normally distributed data and median (range) for those without a normal distribution, and compared using the independent samples t-test and Mann–Whitney U test respectively. All statistical tests were two-sided and p<0.05 was considered as statistically significant.
Results
In the period May 2015 to May 2020, 2318 patients underwent a total of 26,923 intravitreal injections. Ophthalmic nurses and doctors delivered 20,421 (75.8%) and 6502 (24.2%) injections respectively over the five-year period. The annual number of injections delivered by ophthalmic nurses increased year-on-year from 2112 injections in 2015-2016 to 5410 injections in 2019-2020. The annual number of injections and patients in the nursing injection clinic is detailed in Figure 1.
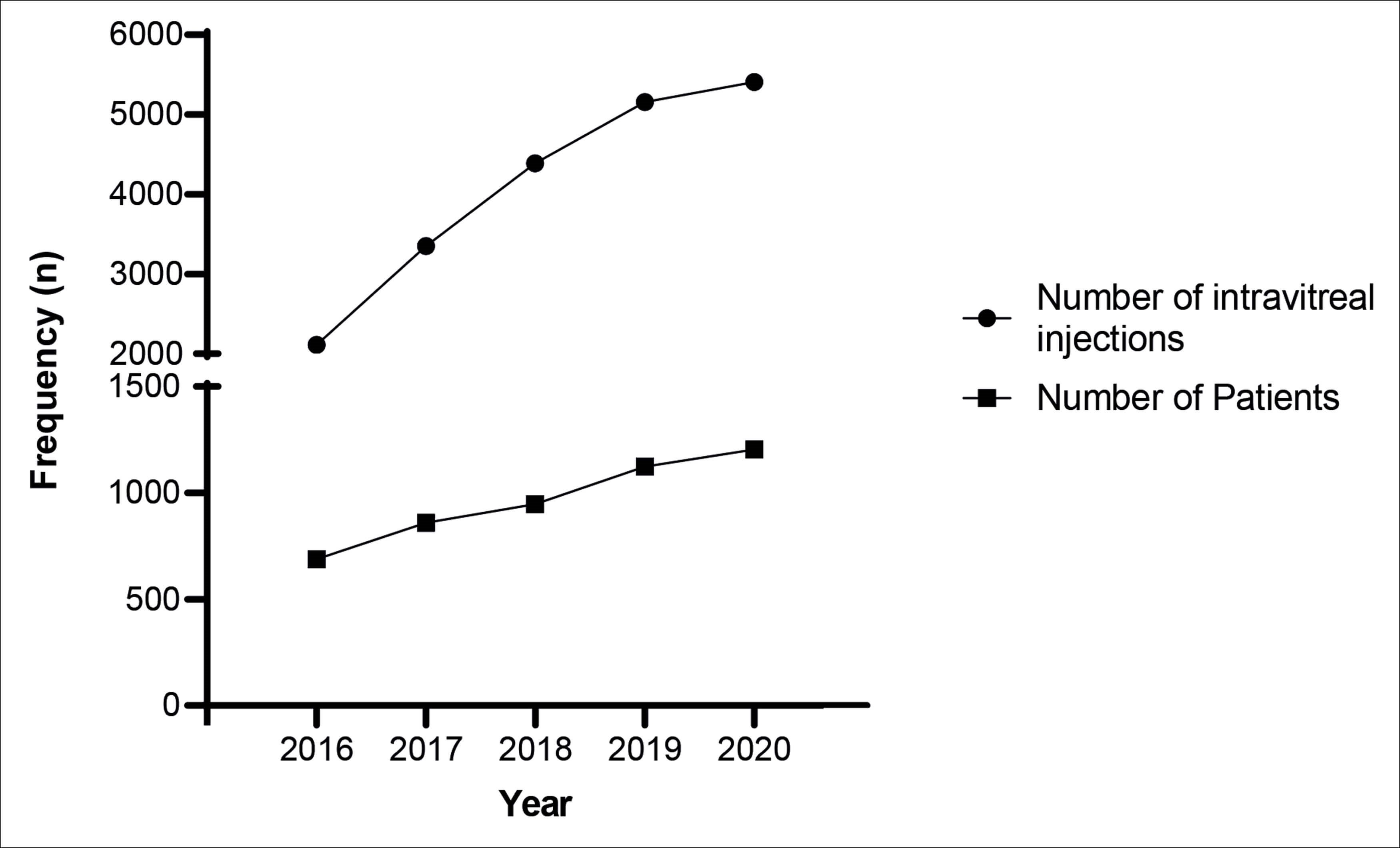
Figure 1: Number of intravitreal injections administered by ophthalmic nurses
and number of patients over the five-year study period.
The number of injections by ophthalmic nurses steadily increased from 2015 to 2020, plateauing in the final year. The annual proportion of injections performed by trained nursing staff increased from 49.9% in year one, peaking to 87.7% by year four. This was reflected by a corresponding decrease in the number of injections by clinicians, which decreased from 51.3% to 12.3% (Figure 2).
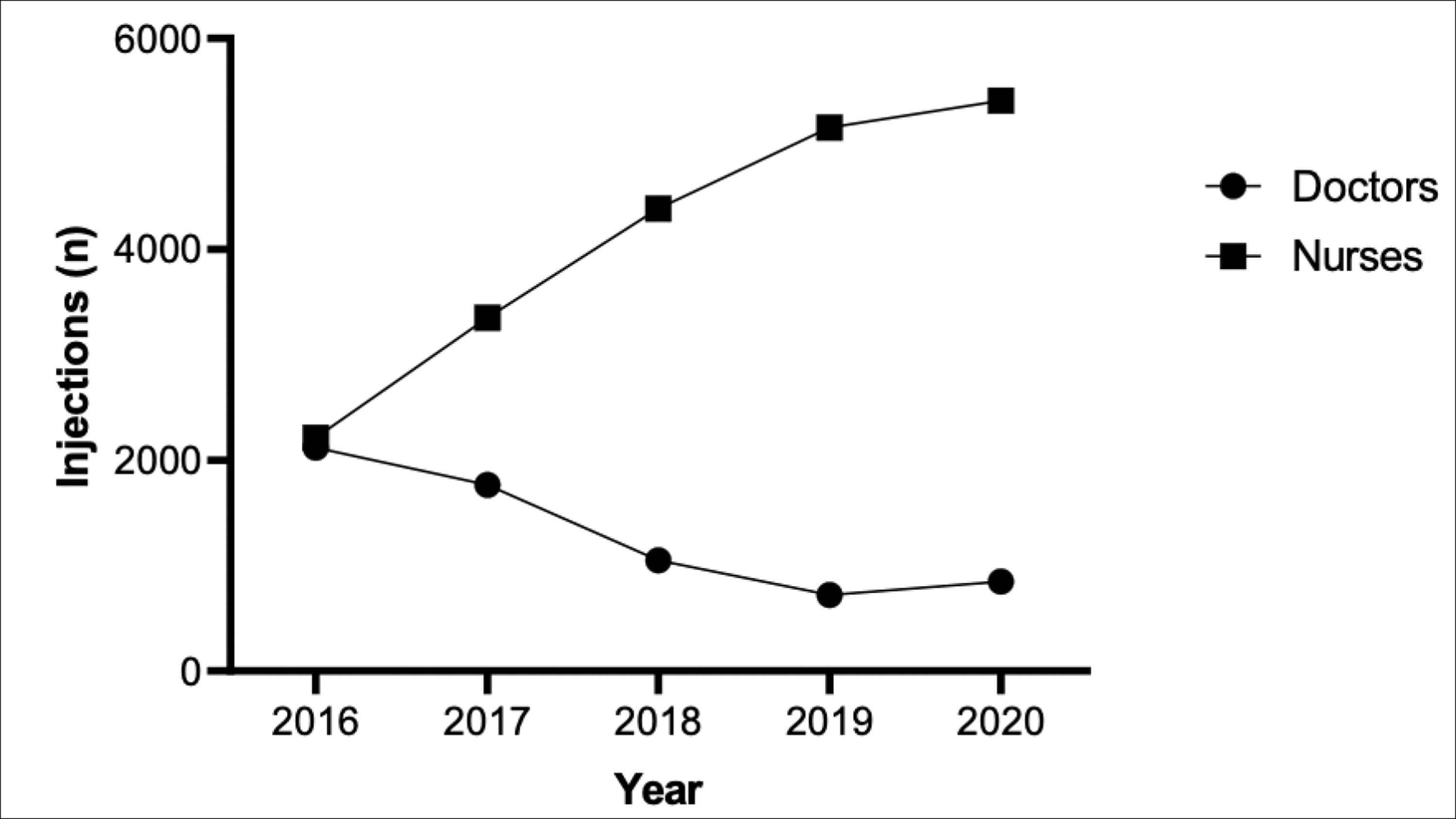
Figure 2: Annual proportion of intravitreal injections administered by ophthalmic nurses vs. doctors.
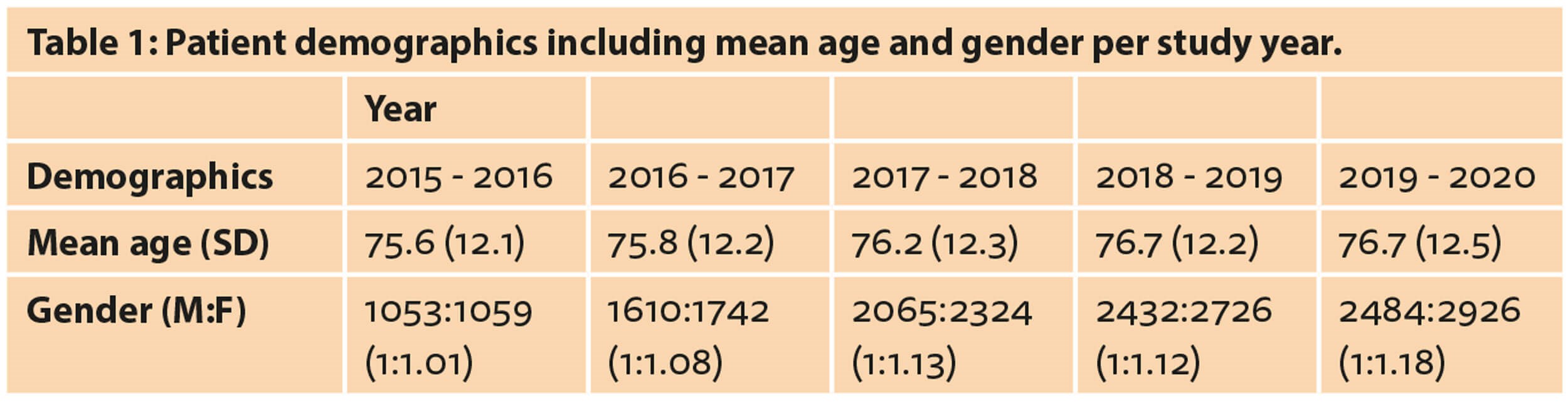
The mean age of patients in 2015 was 75.6 ±12.1 years, increasing to 76.7 ±12.5 years in 2020 (Table 1). A slight female preponderance was noted, with a male to female ratio of 1:1.01 in 2015, increasing to 1:1.18.
Anti-VEGF injections were most commonly indicated for the management of wet age-related macular degeneration (wAMD), with 1174 injections administered in 2016 increasing to 3634 injections in 2020. Other indications for intravitreal injections, in decreasing frequency, included diabetic maculopathy, retinal vein occlusion and retinal angiomatous proliferations. The percentage of intravitreal injections given for wAMD in this study was the greatest, increasing from 55% to 67% by the end of the five-year period. This is in contrast to the percentage of injections provided for diabetic maculopathy and retinal vein occlusion, which decreased from 26% to 19% and 16% to 10% respectively (Figure 3).
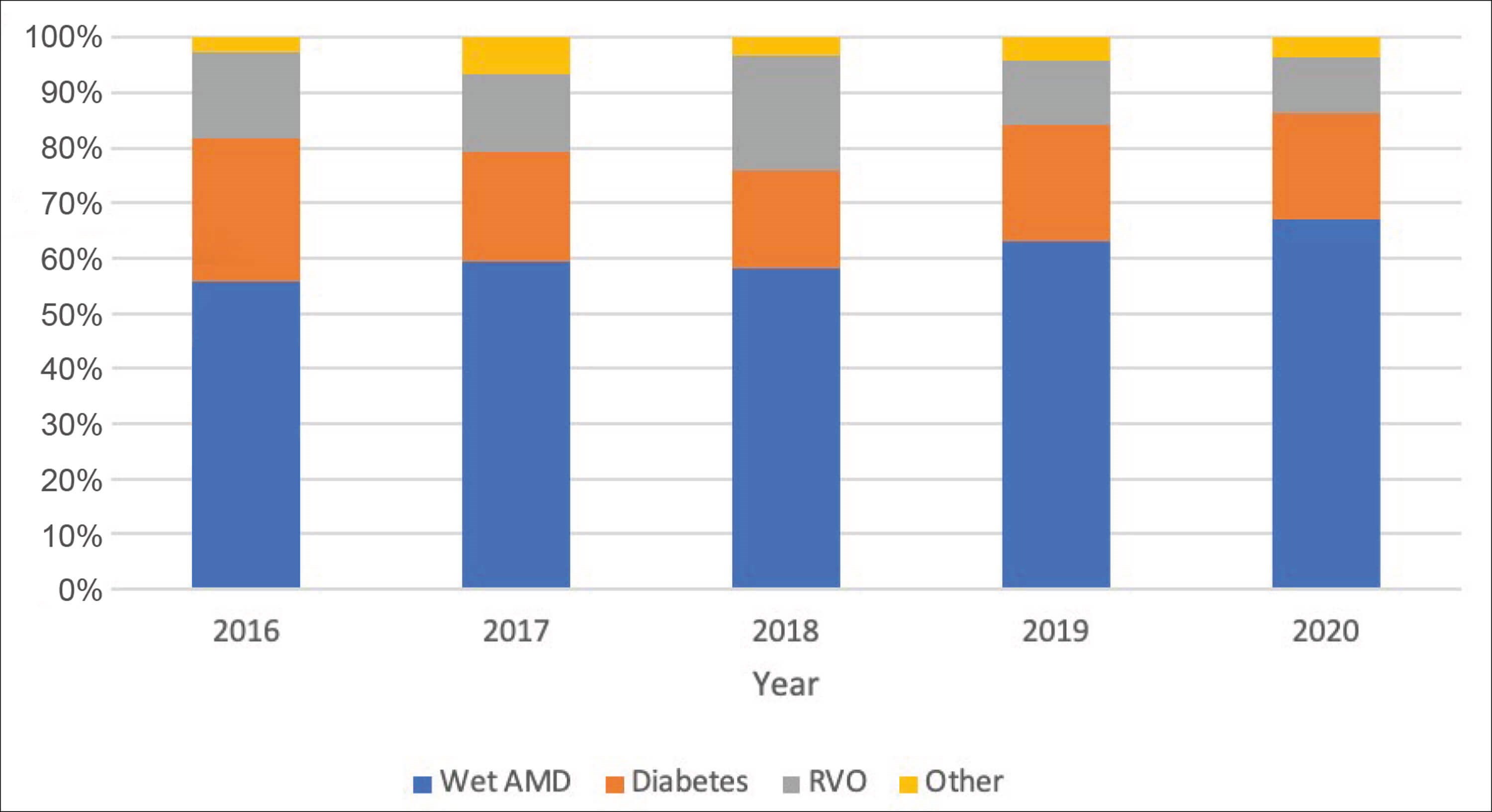
Figure 3: Annual proportion of intravitreal injections administered per indication.
The overall complication rate remained consistently low, ranging from 0% to a peak of 0.137% in year three; this was due to six cases of post-injection intraocular pressure (IOP) spike. These were managed with either topical or oral IOP lowering agents and, in one case, an anterior chamber paracentesis was performed. The rates of post-injection IOP spikes then decreased over years four and five, with rates of 0.058% and 0.056% respectively. There was one case of corneal abrasion (0.030%) and no cases of lens touch, retinal detachment or systemic thromboembolic phenomenon in the data set. Figure 4 outlines the annual percentage of complications.
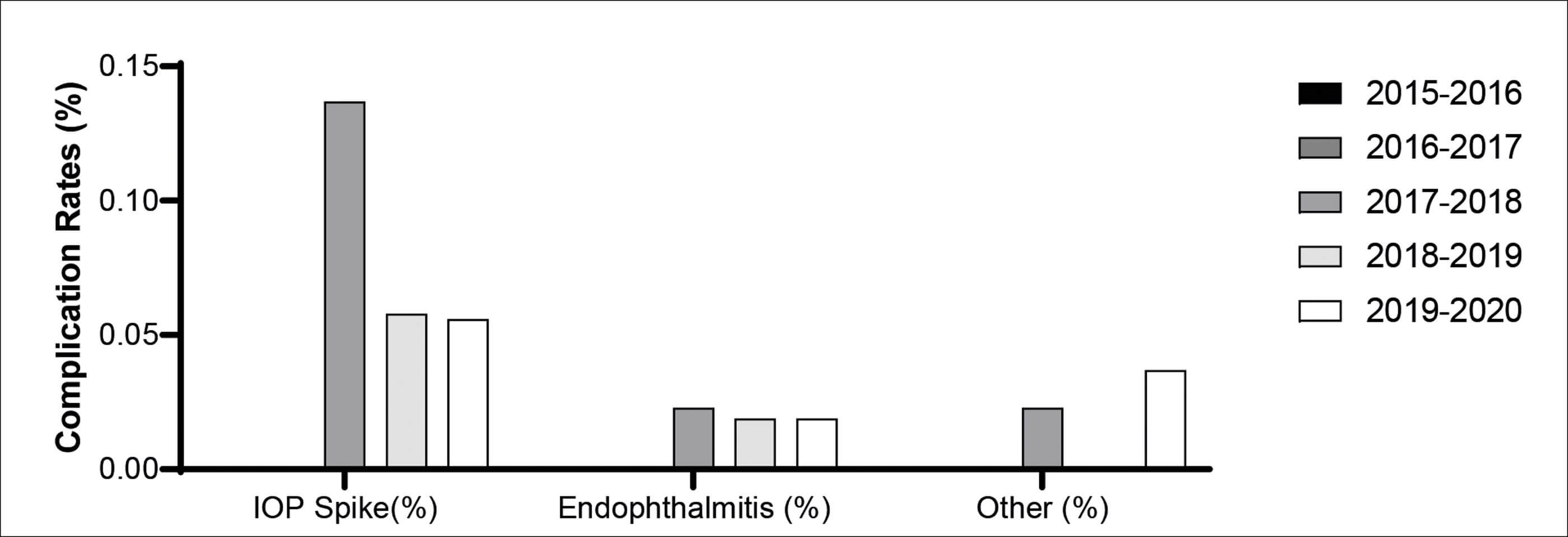
Figure 4: Annual percentage of complication types.
Annual rates of endophthalmitis were as follows: 0% (2016), 0.030% (2017), 0% (2018), 0.039% (2019) and 0% (2020). Three patients over the five-year study period developed a culture negative, post-injection endophthalmitis giving a rate of 0.015%.
Case 1: An 82-year-old male developed endophthalmitis three days after an intravitreal aflibercept injection and was treated with intravitreal vancomycin and ceftazidime. Response was initially poor and the patient underwent pars plana vitrectomy with further intravitreal antibiotics. Visual prognosis remained guarded as the injected eye was previously amblyopic.
Case 2: An 89-year-old female developed endophthalmitis four days following an intravitreal ranibizumab injection and was treated with intravitreal vancomycin and ceftazidime, followed by a pars plana vitrectomy with further intravitreal antibiotics. After 12 months, the patient had a spectacle corrected logMAR visual acuity of 0.58, improving to 0.38 through a pinhole.
Case 3: A 60-year-old male developed a culture negative endophthalmitis five days after an intravitreal aflibercept injection. This was managed with intravitreal vancomycin and ceftazidime. One week later, a pars plana vitrectomy with further intravitreal antibiotics was undertaken. After 12 months, the vision remained poor at hand movements.
Discussion
In the present study, a total of 20,421 intravitreal injections were undertaken over five years by trained ophthalmic nurses using the Precivia® assist device with an endophthalmitis rate of 0.015%, demonstrating safety and efficacy.
Hasler et al. retrospectively reviewed 38,000 intravitreal injections over a three-year-period in Denmark and reported that 63% of injections were performed by nurses with an overall rate of endophthalmitis of 0.36% [6]. In our dataset, 75.8% of all intravitreal injections were administered by ophthalmic allied healthcare professionals. However, this was facilitated by the use of the Precivia® assist device, which has already been shown to be well accepted by ophthalmic nurse injectors as well as patients [7]. Hasler et al. utilised a costlier approach to injecting, consisting of disinfecting the periocular skin with a 5% povidone iodine solution and application of a sterile drape [6]. Our study bypasses the need for this or other instruments, saving expense and time. In a prospective review by Ratnarajan et al., using the assist device in preference to a traditional pack (containing a surgical drape, lid speculum, callipers, disposable Moorfields forceps, two gallipots, gauze and gauze holders, tray cover and needle disposal block) resulted in a saving of £7.70 per patient [8]. Using these figures, for 20,421 patients in the present study, this would save approximately £157,000 over five years, highlighting its economic advantage.
In addition, Hasler et al. reported greater complications in patients with kyphosis owing to poor compliance [6], the device eliminates the issue of difficult positioning as nursing injectors have been able to utilise this with the patient seated semi-recumbent or in Fowler’s position ensuring patient comfort. Contraindications to using the Precivia® include patients with a history of corneal erosions, previous keratoplasty, patients undergone a trabeculectomy or recent phacoemulsification, when the former ‘draping’ method is utilised. Drawbacks of the device include its transparent nature; consequently, the patients’ view remains unobscured, allowing them to see the incoming injection which may heighten anxiety. Further, some injectors find the guided port of the device to be too narrow to facilitate smooth entry with a 30-gauge needle contributing to more patient pain.
The national reported rate of post-injection endophthalmitis rate is 0.025% [9], with rates lower for nurses (0-0.032%) compared to doctors (0-0.042%) [6]. One of the largest safety trials for intravitreal anti-VEGF injections (without use of an assist device) with over 60,000 injections performed had an endophthalmitis rate of 0.02% [10]. The data presented in this study reinforces this, with consistently lower rates of endophthalmitis over the five-year period, suggesting that endophthalmitis rates do not increase if injections are administered by nurses or by using injection assistance devices. Similarly, there were no incidences of lens touch (owing to the design of Precivia®), retinal detachments or systemic thromboembolic events as there has been in other large landmark multicentre trials [11,12].
The department also developed a 12‑week training pathway for nurses from induction to delivering independent intravitreal injections [13]. Between 2015 and 2020, the number of nurse injectors in the department increased from three to seven, while maintaining the standard of injections and keeping patients’ satisfaction at a high level [14]. Not only does this up-skill ophthalmic allied health professionals, but leads to a more proficient nurse-led intravitreal injection service and enables more injections to be undertaken.
Limitations of this study include its retrospective design, but this has enabled a large sample size to be included and, with a greater frequency of new patients commencing treatment than those completing treatment, the study population remained highly dynamic.
Conclusion
The contribution of nursing staff to the administration of intravitreal injections has steadily increased and is expected to increase further in the coming years. Our study demonstrated a marked increase in the proportion of injections performed by nurses between 2015 and 2020 all using the Precivia® assist device, with a high standard of safety and patient satisfaction, highlighting the success of nurse-led injection services in light of an ever-increasing demand for intravitreal injections.
References
1. Hollingworth W, Jones T, Reeves BC, et al. A longitudinal study to assess the frequency and cost of antivascular endothelial therapy, and inequalities in access, in England between 2005 and 2015. BMJ Open 2017;7(10):e018289.
2. Amoaku W, Blakeney S, Freeman M, et al. Action on AMD. Optimising patient management: act now to ensure current and continual delivery of best possible patient care. Eye (Lond) 2012;26 Suppl1(Suppl1):S2-21.
3. Michelotti MM, Abugreen S, Kelly SP, et al. Transformational change: nurses substituting for ophthalmologists for intravitreal injections – a quality-improvement report. Clin Ophthalmol 2014;8:755-61.
4. Simcock P, Kingett B, Mann N, et al. A safety audit of the first 10 000 intravitreal ranibizumab injections performed by nurse practitioners. Eye (Lond) 2014;28(10):1161-4.
5. Varma D, Lunt D, Johnson P, et al. A novel approach to expanding the role of nurses to deliver intravitreal injections for patients with age-related macular degeneration. Int J Ophthalmic Pract 2013;4(2):68-74.
6. Hasler PW, Bloch SB, Villumsen J, et al. Safety study of 38,503 intravitreal ranibizumab injections performed mainly by physicians in training and nurses in a hospital setting. Acta Ophthalmol 2015;93(2):122-5.
7. Hasan H. The role of injection assistant devices in establishing a nurse-led AMD service. Eye News 2018;24(6):14-7.
8. Ratnarajan G, Nath R, Appaswamy S, et al. Intravitreal injections using a novel conjunctival mould: a comparison with a conventional technique. Br J Ophthalmol 2013;97(4):395-7.
9. Lyall DA, Tey A, Foot B, et al. Post-intravitreal anti-VEGF endophthalmitis in the United Kingdom: incidence, features, risk factors, and outcomes. Eye (Lond) 2012;26(12):1517-26.
10. Moshfeghi AA, Rosenfeld PJ, Flynn HW, et al. Endophthalmitis after intravitreal vascular [corrected] endothelial growth factor antagonists: a six-year experience at a university referral center. Retina 2011;31(4):662-8.
11. Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 2006;355(14):1419-31.
12. Brown DM, Kaiser PK, Michels M, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 2006;355(14):1432-44.
13. Hasan H, Mamtora S, Shah N. Setting up a successful nurse-led intravitreal injections service: pearls from Swindon. Br J Nurs 2020;29(20):1178-85.
14. Hasan H, Flockhart S, Qureshi W, et al. Intravitreal injections service: a patient experience evaluation. Br J Nurs 2017;26(12):678-82.
Acknowledgements: The authors would like to thank the eye clinic staff at the Great Western Hospital in Swindon for their support in the completion of this work.
Declaration of competing interests: None declared.
COMMENTS ARE WELCOME