Introduction
The treatments of ocular conditions in the field of ophthalmology varies from topical to surgical procedures. The field of nanotechnology is one of the fast-growing fields of medicine, which plays an important role in turning the impossibilities of the medicinal field into possibilities.
Nanotechnology is concerned with the development and use of materials and technologies on the scale of intracellular structures and molecules, as well as systems and constructions of 100nm in size. The goal of nanotechnology is to provide therapeutic value by full-scale monitoring, regulation, development, restoration, defence, and improvement of human natural systems at the cellular level, using nanoparticles that work a lot in parallel at the single-cell level [1]. Reliable manufacturing techniques and unforeseen biological effects of nanoparticle usage are challenges to nanotechnology. These challenges are not impenetrable, and novel ocular therapies are to be expected in the near future. The other limitations of nanotechnology include higher costs and their complicated development process.
Generations of nanotechnology
The field of nanotechnology has gone through various milestones and generations. The four generations of nanotechnology are [2]:
- Passive nanostructures – first basic generation; includes coatings and particles of nano size.
- Active nanostructures – second generation; can modify certain properties; includes sensors, targeted-drugs, nanomachines etc.
- Nanosystems – nanoparticles of one to 100nm in size; better than conventional systems in delivering drugs efficiently.
- Molecular nanosystems – upgraded generation; can build complex structures based on mechanosynthesis.
Growing difficulties in ophthalmology
Poverty, ignorance, violence, and illness threaten our society. Nations face the issues of a rapidly expanding population, ubiquitous unemployment, increased lifespans, illnesses, insurgencies, and political uncertainty. Despite improvements in healthcare infrastructure and novel treatment methods, ophthalmology and eyecare services demand a quick overhaul. The difficulties in the field of ophthalmology are [3]:
- Blindness and impairments in vision
- Cataracts
- Refractive errors
- Glaucoma
- Macular degeneration
- Trachoma
These problems may be managed using advanced treatment methods available, but a complete cure is not possible, as ophthalmology has the following limitations:
- Lack of specialised healthcare workers
- Limited access towards the residents
- Unequal distribution of the ophthalmologists
Nanotechnology in ophthalmology
“Nanotechnology is an idea that most people simply do not believe” — Rolph Merkle.
The nanoparticles encapsulate the drugs to be delivered in a safe way, which in turn reduces the chances of degradability of drugs and increases their stability, cellular penetration and solubility. The nanoparticles used in the medical field include liposomes, polymers, and surfactants. Nanotechnology may be applied to reduce oxidative stress and treat ocular degeneration, using regenerative medicine to effectively treat ocular conditions.
The most commonly used treatment measures in ophthalmology involving nanoparticles are drug discovery and targeted drug delivery, tissue regeneration and implantable devices, genetics testing, pressure monitoring in eyes, pathophysiology observance of ophthalmic diseases and ocular imaging [4]. Nanotechnology is currently being used in ophthalmology to assess and treat many disease stages (including early and late-stage illness), and many more advances are expected in the coming years. Controlled release of drugs is one of the most important properties of nanotechnology based medicine.
Turning impossibilities into possibilities
“The progression of history is to take the impossible and turn it into the possible.”
Although the applications of nanomedicine and nanoparticle mediated drugs are countless, we have discussed three major subfields using nanoparticles in the field of ophthalmology.
i. Regenerative nanomedicine in ophthalmology
The regenerative nanomedicine involves the cellular and tissue replacement of damaged tissues using the new cells and tissues generated using the advanced methods and materials [5]. Nanoparticles carrying gene transcription factors and other modifying chemicals are used in regenerative nanomedicine, as are nanomaterials to stimulate selective differentiation of neural progenitor cells and to develop neuro-mechanical interfaces. The regenerative nanomedicine has been observed to help us overcome the challenges of ophthalmology [6]. One of the most prevalent ocular conditions worldwide is glaucoma. The impossibilities and limitations in the treatment of glaucoma have been found to be overcome by nanotechnology based treatments. The nanoparticles carrying the drugs, termed nanocarriers, are majorly involved in the intensification of drug delivery. The nanocarriers have been reported to possess the ability to transport to almost all the ocular compartments, which makes their drug delivery more efficient. The nanocarriers are capable of transporting minute to larger drug molecules with or without stability. These nanocarriers loaded with the biological molecules are highly efficient with fewer side-effects than biological molecules without them. They have shown proven effects in the treatment of ocular conditions like glaucoma, occlusion in veins resulting in oedema of eyes, iridocyclitis, ocular tumours etc. Eudragit RS100 (chloro trimethyl-ammonium methyl methacrylate) is a nanoparticle loaded with ibuprofen developed for the treatment of ocular diseases [7].
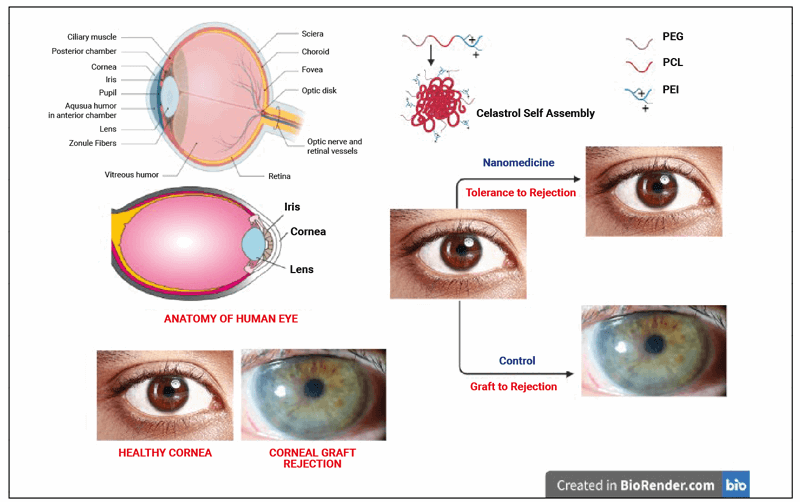
Figure 1: Anatomy of the human eye and cornea before and after allograft corneal rejection.
ii. Corneal allograft rejection and nanoparticles
Corneal allograft rejection may be defined as the immunological response of the host due to antigens in the cornea of the donor. It may destroy the cells of the cornea. Figure 1 shows the healthy and graft-rejected human eye. Nanoparticles have been shown to reduce the corneal allograft rejection by 17% in in vivo studies, when compared to the clinically available drugs. The nanoparticles used were loaded with chitosan [8]. Nanoparticles loaded with rapamycin have been reported to reduce the allograft rejection in corneal transplantation more significantly than conventional rapamycin drugs [9]. The pathophysiology of corneal rejection and the targeted pathways of nano-systems in corneal graft rejection tolerance has been shown in Figure 2.
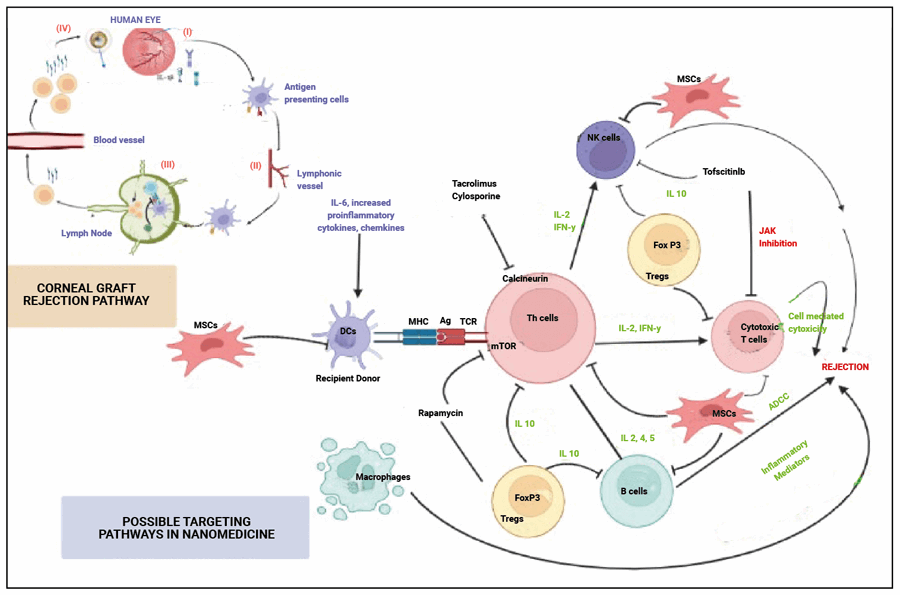
Figure 2: Overcoming corneal graft rejection – nanomedicine targets.
Another study has reported nanoparticles loaded with tacrolimus effectively prevent organ rejection of the cornea during ocular transplantation. The nanomaterial-based drug delivery has hence appeared possible and effective with more advantages than discomfort or side-effects. One of the recently proven nanomedicines in the field of ophthalmology are nanoparticles loaded with celastrol to prevent allograft rejection in corneal transplantation. The toll-like receptor 4 expression as well as macrophage recruitment have been prevented by celastrol in these cases. This helps the patients to overcome the corneal allograft rejection more efficiently than the conventional drugs [10].
iii. Gene therapy and gene delivery in ophthalmology
Ophthalmic gene therapy is the purposeful and smart programming of desired gene expression in certain cells of the eye to treat dystrophies and other genetically based ocular conditions. Nanotechnology can be regarded as a safe and potential tool for the gene delivery and therapy in eyes [11]. The major hereditary conditions of the eyes are retinitis pigmentosa (due to retinal rod cell loss, resulting in tunnel vision and night blindness), Usher syndrome, Stargardt’s disease, Leber congenital amaurosis, choroideremia, diabetic retinopathy, glaucoma, etc. Gene delivery follows two approaches: vector-based and cargo-based, among which vector-based is the most commonly used approach. The nanoparticles used in drug delivery in the eyes have been diagrammatised for easier reference (Figure 3).
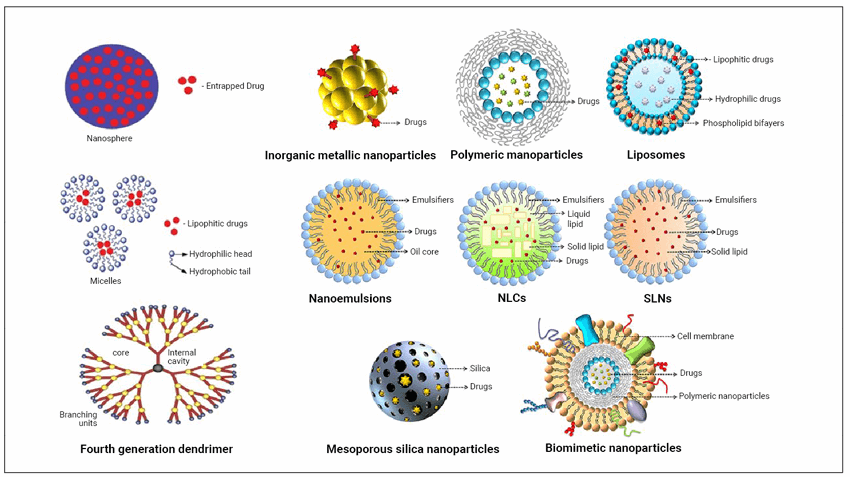
Figure 3: Different types of nanoparticles in drug delivery
(SLNs: solid lipid nanoparticles; NLCs: non-structured lipid carriers).
Nanoparticles: basic science turned into reality
Liposomes are advantageous as nanoparticles due to their degradability, compatibility, non-toxicity, size and their efficiency to encapsulate all forms of drugs [12]. Most used liposome mediated nanoparticles in ocular treatments are as follows: (1) liposome loaded with acyclovir showed higher penetration rate; liposome along with edaravone efficiency managed macular degeneration; latanoprost encapsulated by liposome reduced intraocular pressure. The other nanoparticle mediated treatments are: (2) solid lipid nanoparticles coated with tobramycin for enhanced bioavailability and retention of preocular drugs; (3) polymeric nanoparticles like glycol chitosan cerium oxide increase drug permeability and superoxide dismutase expression in eyes; Eudragit RS 100 polymeric nanoparticles play an important role in controlled release of ocular drugs as well as their retention; triamcinolone acetonide drug when encapsulated by hydroxypropyl-beta-cyclodextrin and polylactic-co-glycolic acid shows enhanced drug delivery; (4)
Polyamidoamine dendrimers are the most commonly used; oligonucleotide encapsulated by dendrimers prevents the progression of choroidal neovascularisation. Nanosuspensions and nano-emulsions are other nano-formulations used in topical ocular treatments [13,14]. As per recent reports, from 2017 to 2020, three new nano-formulations have been approved for human use for different clinical conditions, such as Vyxeos, Onpattro, and Hensify. Visudyne was the last drug approved for macular degeneration in the year 2000 by the Food and Drug Administration [15].
Advantages and limitations of nanomedicine so far
The major advantage of nanomedicine is its safety and bioavailability compared to the corresponding conventional drugs. The nanoparticles can easily penetrate, as well as encapsulate, almost all forms of drugs more efficiently. The permeability of nanoparticles into tumour cells, skin cells, and even the blood-brain barrier has added an advantage to nanomedicine. The other potential advantages are tenability, non-invasive nature, increased rate of dissolution, improved performance, and multifunctionality. More sensitive and efficient detective devices can be developed using nanotechnology, which may detect deadly diseases at an earlier stage, paving the way for better treatment. The limitations of nanomedicine include designing the validation process and devices for nanoparticles, which is much more complicated. The immunogenicity of nanoparticles is still unclear. Although it is safe, the possibility of hypersensitive reactions cannot be ruled out. Almost all the nanoparticles are biodegradable in nature, but nanocarriers that are metallic and non-degradable may be of concern.
Conclusions
In clinical ophthalmology, intrinsic flaws associated with traditional therapeutic procedures often undermine treatment success, pushing the fast development of adaptable alternatives for adequate diagnostics and therapies. Hence, nanomedicine and nanoparticle encapsulated medicines are promising strategies to treat ocular conditions, which have been regarded as impossible and difficult so far. The development of nanomedicine would create a paradigm shift which can revolutionise the whole healthcare field in the coming years. Nano-ophthalmology is a budding field which shows a light of hope in ocular therapy and treatments.
TAKE HOME MESSAGE
-
Nanoparticles are capable of controlled drug release.
-
Corneal allograft rejection can be efficiently prevented by nano-based drugs.
-
Regenerative nanomedicine is significant for intensified drug delivery in ophthalmology.
-
Visudyne was the last nanotechnology drug approved in ophthalmology.
-
Nanoparticles are highly reliable, safe and efficient.
References:
1. Zarbin MA, Montemagno C, Leary JF, Ritch R. Nanotechnology in ophthalmology. Can J Ophthalmol 2010;45(5):457-76.
2. Roco MC. The long view of nanotechnology development: the National Nanotechnology Initiative at 10 years. J Nanoparticle Res 2011;13(2):427-45.
3. Heidary F, Gharebaghi R. Significance of novel ideas to solve challenges facing today’s ophthalmology. Med hypothesis, Discov Innov Ophthalmol J 2012;1(2):19-20.
4. Petrovic N, Petrovic MJ, Sreckovic S, et al. Nanotechnology in ophthalmology. In: Commercialization of Nanotechnologies–A Case Study Approach. New York, USA; Springer International Publishing; 2018:275-97.
5. Zarbin MA, Montemagno C, Leary JF, Ritch R. Regenerative nanomedicine and the treatment of degenerative retinal diseases. WIREs Nanomedicine and Nanobiotechnology 2012;4(1):113-37.
6. Stern JH, Tian Y, Funderburgh J, et al. Regenerating eye tissues to preserve and restore vision. Cell Stem Cell 2018;22(6):834-49.
7. Brabazon D, Pellicer E, Zivic F, et al. Commercialization of Nanotechnologies–A Case Study Approach. New York, USA; Springer International Publishing; 2018.
8. Yuan X-B, Yuan Y-B, Jiang W, et al. Preparation of rapamycin-loaded chitosan/PLA nanoparticles for immunosuppression in corneal transplantation. Int J Pharm 2008;349(1-2):241-8.
9. Wei C, Wang Y, Ma L, et al. Rapamycin nano-micelle ophthalmic solution reduces corneal allograft rejection by potentiating myeloid-derived suppressor cells’ function. Front Immunol 2018;9:2283.
10. Li Z, Liu R, Guo Z, et al. Celastrol-based nanomedicine promotes corneal allograft survival. J Nanobiotechnology 2021;19(1):341.
11. Mitra RN, Zheng M, Han Z. Nanoparticle‐motivated gene delivery for ophthalmic application. WIREs Nanomedicine and Nanobiotechnology 2016;8(1):160-74.
12. Rehman AU, Akram S, Seralin A, et al. Lipid nanocarriers: Formulation, properties, and applications. In: Nguyen-Tri P, Do T-O and Nguyen TA, (Eds.). Smart Nanocontainers. Amsterdam, Netherlands; Elsevier; 2019:355-82.
13. Li F, Wen Y, Zhang Y, et al. Characterisation of 2-HP-β-cyclodextrin-PLGA nanoparticle complexes for potential use as ocular drug delivery vehicles. Artif Cells, Nanomedicine, Biotechnol 2019;47(1):4097-108.
14. Khiev D, Mohamed ZA, Vichare R, et al. Emerging nano-formulations and nanomedicines applications for ocular drug delivery. Nanomaterials 2021;11(1):173.
15. Anselmo AC, Mitragotri S. Nanoparticles in the clinic: An update. Bioeng Transl Med 2019;4(3):e10143.
Declaration of competing interests: none declared.
COMMENTS ARE WELCOME








