Safa Elhassan gives a brief review of amniotic membrane grafts and their application in theatre and clinic-based settings.
Amniotic membrane (AM) transplant has been an established adjuvant treatment for many corneal, conjunctival and scleral disorders in ophthalmic clinical practice with successful outcomes. It is therefore imperative for all trainee ophthalmologists to understand the properties, its application, as well as the new forms of sutureless amniotic membrane graft (AMG).
“Amniotic membrane as a graft is placed epithelial side UP (sticky side touching host’s cornea). Amniotic membrane as a patch is placed epithelial side DOWN (smooth and shiny surface of the membrane touching host’s cornea), though it can be placed epithelial side UP too”
Amniotic membrane
The amnion is the inner membrane that makes up the amniotic sac. This avascular structure initially surrounds the embryo and then, as the foetus grows in size, is exposed to amniotic fluid. The chorion is the vascular outer membrane that is in contact with the uterine wall and makes up the rest of the amniotic sac [1].
The amnion is a thin tissue measuring 0.02-0.05mm in thickness and has three different layers:
- Epithelium
- Basement membrane
- Stroma
The epithelium is a single cuboidal or columnar cell layer with surface microvilli, which project into the amniotic fluid [1].
The basement membrane is made up of reticular fibres. Although thin, it is one of the thickest basement membranes found in the human body and can with withstand cryopreservation [1].
The stroma is comprised of three layers – compact, fibroblast and spongy. The compact layer is the strongest layer of amnion and can withstand inflammation and oedema. The fibroblast layer is thick and made up of fibroblasts within reticular tissue which occasional display phagocytic potential. The outer most layer is spongy gelatinous-like and made up of reticulin bundles in mucin. This layer can become oedematous and allows the amnion to slide over the securely tethered chorion [1].
One placenta can provide up to 40 amniotic membrane grafts for ophthalmic use [2].
Amniotic membrane preparation
The donor placentas are obtained from maternal donors during elective caesarean section. All maternal donors are screened with appropriate social and medical history; consented and tested for hepatitis B & C, syphilis, HIV and additional serology before selection. This complex process is carried out in line with multiple EU and UK standards and guidelines [2].
The amnion is dissected from the chorion in clean rooms and bathed in a 24 hour ‘cocktail’ of antibiotics and washed with balanced salt solution (BSS). The AM is held for six months to allow maternal donors to be re-tested before it is released [2].
Amniotic membrane can be preserved through cryopreservation (CHAM) or in a dry de-epithelialisation form (DHAM) [2].
Cryopreserved human amniotic membrane (CHAM)
The AM is cut into pieces of small (2cm by 2cm) or large (3cm by 3cm) sizes and placed on nitrocellulose paper (stromal side down). The AM is stored in solution of 50% glycerol and frozen at temperatures of -80 degrees [2]. CHAM can be kept for 48 hours and must be stored in a freezer below -40 degrees. Beyond this time a license is required from the Human Tissues Authority (HTA). CHAM is thawed 10 minutes before use at room temperature and must be used immediately [1,2].
Dry human amniotic membrane (DHAM)
The AM is sterilised in a similar way to cryopreserved AM but preserved using low heat and air vacuum. Some DHAM is sterilised using low energy electron beam radiation. As it is dry it does not require placement on nitrocellulose paper. It can be stored at room temperature and kept for two to five years (HTA license required for storage beyond 48 hours). DHAM is usually hydrated before use [1,2].
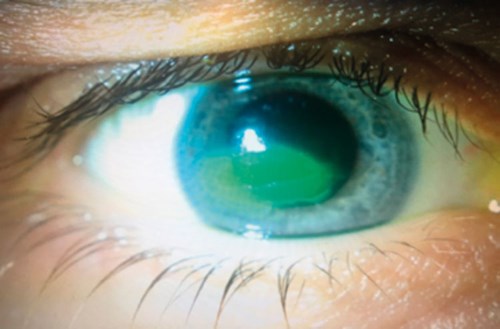
Figure 1: Persistent epithelial defect that failed with conventional management.
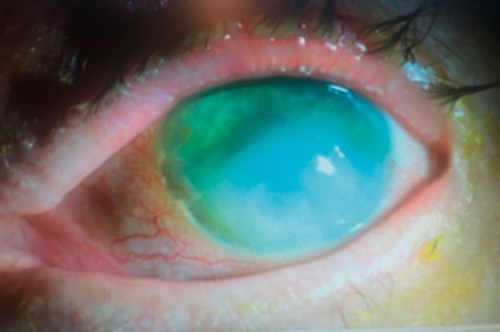
Figure 2: Stenotrophomonas and Elizabeth Kingia miricola keratitis
after the use of Omnigen for neurotrophic persistent epithelial defect (PED).
When is an amniotic membrane used?
AM can be used as a patch to promote re-epithelialisation, or as a graft which acts as a substrate and a scaffold for epithelial growth [1]. It has been used in a wide range of ophthalmic conditions. The Table 1 details some of the uses for AM (see below)
Complications of amniotic membrane use
- Risk of transmission of bacterial, viral or fungal infections to the recipient [1]
- If donors are not adequately screened for communicable diseases
- If AM is not processed under sterile conditions
- If AM is not stored appropriately. - Post AM transplantation infection, particularly with gram positive bacteria [1]. (See Figure 2 – a post Omnigen insertion microbial keratitis).
- Premature degeneration and cheese wiring requiring frequent repeat transplantations [3].
- Persistent subepithelial membrane and inadvertent corneal opacification [3,5].
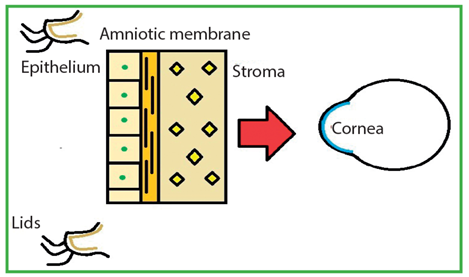
Figure 3: Epithelial side UP amniotic membrane.
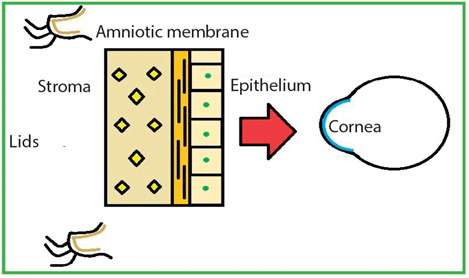
Figure 4: Epithelial side DOWN amniotic membrane.
Amniotic membrane orientation
The orientation of the AM is important and dependent on the endpoint objective.
Orientating the AM epithelial side up (epithelium facing hosts’ lids and stromal AM towards host’s cornea) helps provide the corneal and conjunctival epithelial cells a substrate on which they can grow, therefore aiding re-epithelisation of the host’s epithelium (Figure 3). If placing the AM epithelial side down (epithelium facing host’s cornea and stromal AM towards host’s lids) the stromal matrix helps reduce inflammation (Figure 4) [1,5].
The epithelial side of AM is smooth and shiny, whilst the stromal side of AM is sticky and gelatinous. It is provided on nitrocellulose filter paper usually with epithelial side up and stromal down on the paper [2].
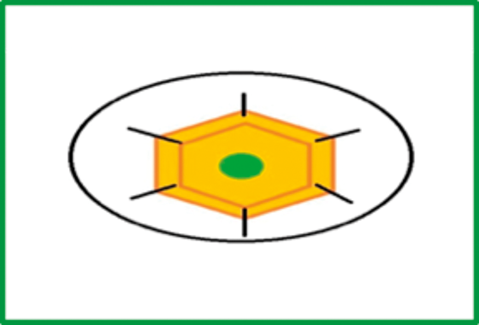
Figure 5: Diagram of amniotic membrane graft. The corneal defect (green circle) is
covered by the amniotic membrane (orange hexagon) held with interrupted corneal sutures.
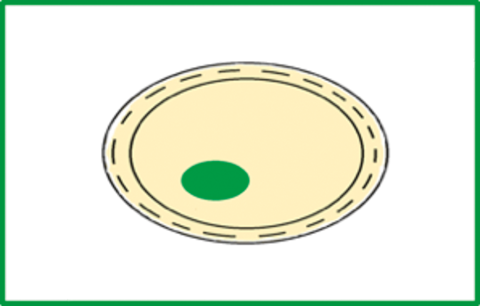
Figure 6: Diagram of amniotic membrane patch. The corneal defect (green circle)
is covered with amniotic membrane patch (cream circle) sutured in place with
vicryl in a purse-string fashion in the peri-limbal region.
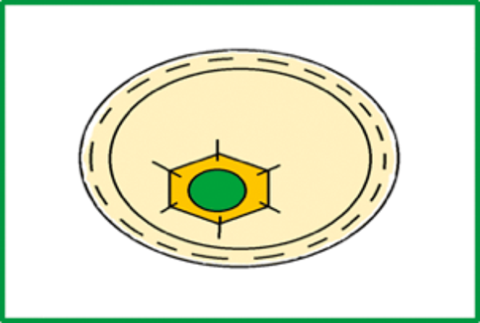
Figure 7: Diagram of combined AM. The corneal defect (green circle) is filled
with AM graft (orange hexagon) and then covered with AM patch (cream circle).
Sutured AM in a theatre based setting
Types
Graft or inlay (permanent basement membrane substrate or scaffold)
The AM is orientated with the epithelial side up and trimmed to an appropriate size or folded in place. About 2mm of epithelium surrounding the wound is debrided. The AMG is secured to the cornea to the episclera and conjunctiva with 10-0 vicryl (Figure 5). The host epithelial cells regenerate and grow over the basement membrane of AM. Stromal AM therefore may become integrated into host cornea, hence the term ‘graft’. Single layer or multiple layers of AM can be applied depending on the size of the defect [1,6].
Patch or overlay (temporary basement membrane)
AM is used either epithelial side up or down. It is sutured over the host’s epithelium to conjunctiva or episclera using 10-0 vicryl (Figure 6) or glued in place with fibrin. In this way AM acts like a ‘natural’ patch and usually breaks down after one to two weeks [1,6].
Combined (inlay and overlay)
This is a combined technique in which the graft covers the host defect and the patch is place over both creating a ‘sandwich’ effect (Figure 7). This allows the epithelium to grow under the patch and above the graft [1,6].
Application of AM as a patch or graft
Preparation: Operating theatre, microscope, iodine surgical solution, surgical drape, topical or local anaesthetics, eyelid speculum, surgical scissors, needle holder, tying forceps, toothed forceps, non-toothed forceps, calliper, bandage contact lens (BCL) sizes 16-20mm, BSS, surgical gauze, sutures: nylon 11-0 or10-0 and vicryl 10-0 – 9-0 [6].
- Perform the appropriate anaesthetic block such as sub-tenons or peribulbar.
- Clean, drape and place speculum onto the eyelids and eye in a similar fashion as one would for cataract surgery preparation.
- Remove the AM from the nitrocellulose paper by using two forceps and grabbing the two corners while the assistant peels the paper away (stromal / sticky side is usually adherent to the paper but check with a surgical sponge).
- a) Graft: for small full or partial thickness stromal defects. The AM can be cut into small pieces to layer or fill the defect (orientation not important) and then a larger AMG is place on top – epithelial side up. Suture the AMG tightly over the underlay with 10-0 nylon interrupted or continuous 10-0 nylon sutures [6] (Figure 5).
OR
b) Patch: Unfold and spread the AMG – epithelial side down or up over the corneal surface until it is a single layer. Secure the AM graft using 10-0 vicryl and suture 2-3mm parallel to the limbus in a purse string fashion with 8 to 10 episcleral bites [6]. Start at the 6 or 12 o’clock location and end the final knot unburied. - Cut excess AMG or fold and suture the ends together.
- Place a BCL on top.
- Consider temporary tarsorraphy.
Sutureless AM in a clinic-based setting
Sutureless AM (SLAM) have become more commercially available and can be used outside the theatre setting without the need of suturing the AM in place.
Types
SLAM come either in cryopreserved or dried form with their own bandage contact lens or self-retaining bandage device. Generally, each commercial company obtains the AM in a similar manner to the processes described above but with slight variation in its preparation, such as different antibiotics used in the ‘antibiotic cocktail wash’ [7].
Similar to CHAM, cryopreserved sutureless AM (CSLAM) must be stored at temperatures below -80 degrees and thawed to room temperature before use. Dried sutureless AM (DSLAM) can be kept at room temperature and rehydrated before used. Both can be stored for up to two years with a HTA license.
Table 1: The different uses of AM in graft or patch form with different structures [1,3-5].
Cornea
Graft: Small perforations or melts secondary to corneal ulcers or thinning (sterile)
Patch: Persistent epithelial defects (Figure 1); Neurotrophic keratitis; Band keratopathy, Bullous keratopathy; Limbal stem cell disease; Recurrent epithelial erosion; Corneal dystrophy
Conjunctiva
Graft: Pterygium excision; Bleb reconstruction; Symblepharon; Reconstruction of the conjunctiva and fornix following tumour excision or cicatrising disease
Patch: Symblepharon; Mechanical trauma; Chemical trauma; Stevens-Johnson syndrome
Scleral
Graft: Small scleral perforations and melts
Patch: Large melts or perforations
Others
Graft: Eyelid reconstruction
Patch: Glaucoma or cataract surgery; Dysfunctional tear syndrome
Use and complications
SLAM can be used for most diseases noted in Table 1 but may not be suitable for peripheral corneal disease, active infection, perforations, large defects and lid reconstruction [5]. They are also contraindicated in patients who have had glaucoma drainage devices or filtration surgery. Caution should be taken in patients with sensitivity to antibiotics or chemicals used in the preparation of SLAMs [7,8]. In addition to the complications mentioned above, discomfort, intolerance, BCL slippage or displacement, blurred vision and membranes not dissolving have all been reported with SLAM [5].
Method and orientation
Each company provides a comprehensive guideline for SLAM insertion. CSLAM usually comes in one orientation – epithelial side up, whilst DSLAM has both options of epithelial side up or down, which is marked.
CSLAM – ProKera® (Bio-tissue) [7]
Insertion:
Preparation: Gloves, sterile saline solution, sterile cotton buds, anaesthetic eye drops, sterile gauze, gallipot and surgical tape.
- Allow the product to thaw to room temperature (about nine to ten minutes).
- Carefully open the packaging and drain the excess storage solution around the ProKera held in a clip in a gallipot or sink.
- Rinse the ProKera twice using sterile saline whilst in its storage container.
- Place topical anaesthetic into the patient’s eye.
- Wash your hands and place on sterile gloves. Then remove the ProKera from the holding clip whilst in the storage container
- Ask the patient to look down and insert the ProKera lens into the upper lid fornix. Then ask the patient to look straight ahead to ensure it is inserted into the lower lid fornix.
- Check at the slit-lamp and document appropriately with correct labels.
Removal:
Preparation: Gloves, sterile forceps, anaesthetic eye drops, sterile gauze and sterile cotton buds.
- Place anaesthetic drops in the eye.
- Ask the patient to look up and use the forceps to pinch the lower edge of ProKera lens and gently pull out the lens.
- Don’t leave Prokera for longer than 30 days on the eye.
DSLAM – Omnigen® (NuVision) [8]
Insertion:
Preparation: Gloves, sterile saline solution, anaesthetic eye drops, sterile gauze and sterile non-toothed forceps.
- Place anaesthetic drops into patients affected eye.
- Remove the outer packaging and then open the sandwiched sterile trays containing the Omnigen.
- Open the OmniLenz (bandage contact lens) and either pour out the excess fluid or use non-toothed forceps to remove the lens and dry with the surgical spears provided.
- Place the Omnigen into the contact lens according to your required orientation (place eye logo at 12 o’clock position – eye looking clockwise = epithelial side UP and eye directed anti-clockwise = epithelial side down).
- Allow the Omnigen to rehydrate in the lens for two minutes before inserting onto the patient’s cornea with non-toothed forceps or fingers
- Tilt the patient’s head back and hold both eyelids apart. Ask the patient to look straight ahead and place centrally on the cornea.
- Check position at the slit-lamp and adjust with sterile cotton buds.
Removal:
Preparation: Gloves, sterile forceps, anaesthetic eye drops, sterile gauze and sterile cotton buds.
- Place anaesthetic drops in the eye.
- Ask the patient to look up and use the forceps to pinch the lower edge of OmniLenz and Omnigen and gently pull out.
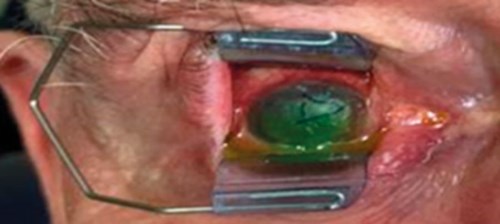
Figure 8: Bladed speculum and an Omnigen. Inserting the Omnigen directly
on the cornea and allow it to rehydrate. Then apply the OmniLenz on top of the Omnigen.
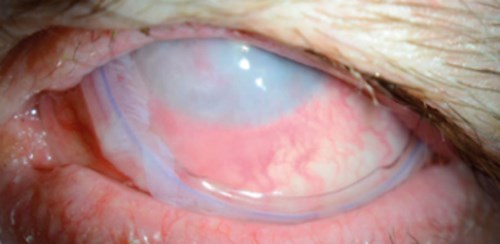
Figure 9: ProKera inserted over a microbial corneal ulceration with a
small perforation, secondary to a vitamin A deficiency.
Practical tips
In clinical practice I find it more convenient using a bladed speculum to keep the eye open while inserting both ProKera and Omnigen (Figure 8). I also find it useful to place the Omnigen directly on to the cornea and then allowing it to rehydrate before applying the Omnilenz (Figure 8). Personally I would not use Omnigen or ProKera in active microbial keratitis, but only to aid re-epithelialisation once it is sterile (Figure 9).
I recommend co-ordinating with your theatre or clinic to order the AM in good time. Ensure there is appropriate storage as the AMG can be only be kept up to 48 hours without HTA license. Consent the patient with the risks and review them early to monitor any complications. Preservative free eye drops are preferred after the application of AM. Document clearly the full history, examination and visual acuity. The batch identification label and transplantation document should also be included in the notes. Consider performing tarsorraphy or use botulinum toxin to induce ptosis after inserting the AMG to facilitate corneal healing.
Conclusion
AM is useful in treating many corneal disorders. With sutureless AMG more readily available it is important to understand when they are indicated, how to apply and remove these sight-saving devices. The differences between sutured and sutureless amniotic membrane transplants are summarised in Table 2.
Table 2: Summarising the differences between sutured and sutureless amniotic membrane transplant.
Amniotic membrane transplant
Sutured AM – fresh-frozen or freeze-dried
Indications: Superficial and deeper ocular surface disease; perforations; conjunctiva; sclera; glaucoma surgery and others
Contraindications: None
Attachment: Sutured / Glued / BCL
Location: Theatre
Cost: Moderate (AM only) / Expensive (with theatre time and costs)
Time of procedure: Long (45-60 mins)
Storage: Freezer -80C (frozen AM) / Dry 25C (Dehydrated AM)
Shelf-life: Two years
Technique: Challenging / Requires experience
Amniotic membrane transplant
Sutureless AM – Prokera® or Omnigen®
Indications: Superficial and central ocular surface disease
Contraindications: Glaucoma drainage surgery or filtering bleb and antibiotic sensitivity reactions
Attachment: BCL or self-retaining bandage device
Location: Clinic
Cost: Relatively inexpensive
Time of procedure: Quick (five mins)
Storage: Freezer -80C (ProKera®) / Dry 25C (Omnigen®)
Shelf-life: Two years
Technique: Simple / No experience required
References
1. Malhotra C, Jain AK. Human amniotic membrane transplantation: Different modalities of its use in ophthalmology. World J Transplant 2014;24(2):111‑21.
2. NHS Blood and transplant. Amniotic membrane details and specifications; Available at:
https://www.nhsbt.nhs.uk/
tissue-and-eye-services/
products/eyes/amniotic-membrane/
3. Rahman I, Said DG, Maharajan VS, Dua HS. Amniotic membrane in ophthalmology: Indications and limitations. Eye 2009;23(10):1954‑61.
4. Stephenson M. Treatment Options with Amniotic Membrane. Review of Ophthalmology 2015.
https://www.reviewofophthalmology.com/
article/treatment-options-with-amniotic-membrane
5. Meller D, Pauklin M, Thomasen H, et al. Amniotic membrane transplantation in the human eye. Dtsch Arztebl Int 2011;108(14):243-8.
6. Tseng S, Elizondo A, Casas V: Amniotic Membrane Suturing Techniques. Ophthalmic Microsurgical Suturing Techniques. In Ophthalmic Microsurgical Suturing Techniques. Macsai MS (ed). Springer-Verlag Berlin Heidelberg; 2007:107-16.
7. Prokera. Bio-tissue.
https://www.biotissue.com/
products/prokera.aspx
8. Omnigen. NuVision Biotherapies Limited.
http://www.nu-vision.co.uk/omnigen
(All links last accessed April 2019).
TAKE HOME MESSAGE
-
Amniotic membrane can be used as a patch to promote re-epithelialisation, or as a graft, which acts as a substrate and a scaffold for epithelial growth.
-
AM can be used for various conjunctival, corneal or scleral diseases.
-
The epithelial side of AM is smooth and shiny, whilst the stromal side of AM is sticky and gelatinous.
-
Orientating the AM epithelial side up (stromal AM towards host’s cornea) helps provide the corneal and conjunctival epithelial cells a substrate on which they can grow to aid re-epithelisation.
-
Placing the AM epithelial side down (epithelium facing host’s cornea) the stromal matrix helps reduce inflammation.
-
There are various sutureless amniotic membrane devices available, which can be inserted in a clinic-based setting.
-
Patient should be consented with potential risks and complications of having an AMG. Batch identification labels and transplant documentation should also be included in the notes.
Declaration of competing interests: None declared.
COMMENTS ARE WELCOME






