Although several excellent modalities are available for correcting high myopia, the surgical treatment of this condition remains one of the biggest challenges for refractive surgeons; this group of patients is often very dependent on contact lenses. If these patients become intolerant of contact lenses then they will seek out surgical options as an alternative method of vision correction.
Selecting the safest and most appropriate technique for each patient is critical. Today’s refractive surgeons can treat higher levels of myopia more safely and predictably thanks to a better understanding of iatrogenic ectasia, as well as the availability of screening modalities, improved platforms for laser-assisted in situ keratomileusis (LASIK), and better techniques for advanced surface ablation. More advanced intraocular lenses (IOLs) and technology for sizing and delivering these devices have allowed ophthalmologists to use intraocular solutions to maximise visual quality in highly myopic eyes when laser treatment is not appropriate.
Treatment of refractive error can provide patients with considerable benefits. The World Health Organization has listed myopia and uncorrected refractive error among the leading causes of blindness and visual impairment in the world. The prevalence of myopia in Western populations is estimated to be approximately 25% [1]. Myopia can be broadly classified into two groups: low to moderate myopia, which is 7.00D or less of myopic spherical equivalent with or without astigmatism, and high myopia, which is >7.00D of myopic spherical equivalent with or without astigmatism.
Patients with high myopia who have poor vision with spectacles and are intolerant of contact lenses now have several choices for surgical correction. In recent years, ophthalmologists have favoured LASIK and advanced surface ablation for the surgical correction of refractive error in most patients who wish to be independent of spectacles. These procedures provide rapid visual recovery, excellent visual outcomes, and a relatively painless postoperative recovery. For patients with high degrees of myopia, refractive surgery with an excimer laser may be less predictable than treatments for lower levels of myopia. Haze has been reported to be a significant long-term problem in eyes with high myopia treated with photorefractive keratectomy (PRK) [2]. Refractive lens exchange (RLE) may increase the risk of retinal detachment [3], and generally should not be considered in pre-presbyopic patients with myopia who can still accommodate.

Figure 1: Visian implantable collamer lens.
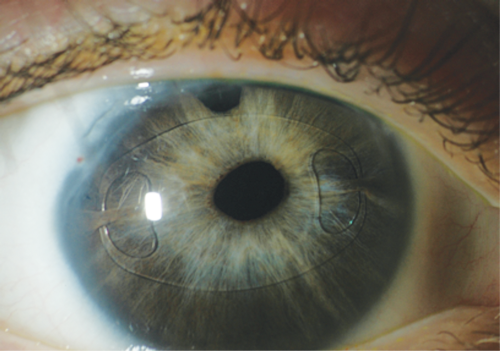
Phakic IOLs represent an alternative surgical treatment for moderate to high myopia. In 2004, the Food & Drug Administration (FDA) approved the Verisyse phakic IOL, marketed internationally as the Artisan lens by Ophtec BV and distributed in the United States by Abbott Medical Optics Incorporated. The Verisyse / Artisan phakic IOL is an iris claw-fixated anterior chamber (AC) IOL. The FDA approved the Visian ICL (implantable collamer lens – also known as the implantable contact lens), manufactured by STAAR Surgical Company, in December 2005 (see Figure 1). The Visian Toric ICL (STAAR Surgical Company) has a toric anterior surface and is designed to vault anteriorly to the crystalline lens in the ciliary sulcus. Phakic IOL implantation has the benefit of being a reversible procedure. Their insertion requires intraocular surgery, and the associated risks include: endophthalmitis, surgically induced astigmatism, loss of corneal endothelial cells, chronic uveitis, pupillary block glaucoma, pigment dispersion syndrome and cataract formation. In addition, the lens power calculation and surgical implantation of phakic IOLs require special techniques, and the long-term outcomes of several types of phakic IOLs are unknown. An iris fixated AC IOL is shown in Figure 2.
Figure 2: Iris fixated AC IOL.
Anterior chamber phakic IOLs
Akcay et al. evaluated two foldable AC IOLs for high myopia in a prospective, interventional case series. The investigators implanted the Artiflex IOL (Ophtec BV), an iris-claw lens, in 62 eyes and the ICare phakic IOL (Cornéal), an angle-supported lens, in 42 eyes. The range of myopia treated was -7.75D to -26.00D spherical equivalent. At the 18-month follow-up, uncorrected visual acuity (UCVA) improved from 1.60 ± 0.10 logMAR to 0.37 ± 0.23 logMAR in the Artiflex group and from 0.70 ± 0.20 logMAR to 0.47 ± 0.14 logMAR in the ICare group. There was a significant reduction in endothelial cell counts in both groups, however, longer term follow-up would be required to ascertain whether this could have any effect on corneal clarity long-term [4].
In a non-randomised, multicentre clinical trial, Lane and Waycaster assessed the impact of bilaterally implanting the AcrySof Cachet phakic IOL (Alcon Laboratories, Inc.) on vision and quality of life in 138 patients with high myopia. Mean uncorrected distance visual acuity at six months, one year, and two years postoperatively was statistically better than corrected distance visual acuity at baseline (-0.12 logMAR, -0.11 logMAR, -0.12 logMAR, respectively, versus -0.06 logMAR). The increase in patients’ satisfaction with their UCVA postoperatively compared with preoperatively was significant, and patients’ distance vision without spectacles improved from 0% preoperatively to 94% postoperatively. The rate of endothelial cell loss was consistent with normal age-related changes in the cornea. Also, most patients reported improved satisfaction with their UCVA and quality of life [5].
Posterior chamber phakic IOLs
In a large cohort study, Alfonso et al. evaluated the long-term safety and efficacy of the Visian ICL for the treatment of high myopia in 188 eyes. The mean spherical equivalent decreased from -11.17 ±3.40 preoperatively to -0.88 ±0.72D five years postoperatively. The mean uncorrected and corrected distance visual acuities (Snellen decimal) were 0.69 ±0.26 and 0.83 ±0.15; this approximates to a Snellen equivalent of 6/9 and 6/7.5, respectively. None of the eyes lost more than two lines of visual acuity, and 70% of eyes achieved 0.80 or better distance best-corrected visual acuity (BCVA). Three eyes (1.6%) developed a late anterior subcapsular cataract, which was clinically significant in one case and required explantation of the phakic IOL and phacoemulsification. Three eyes (1.6%) had a mild, transient increase in IOP (up to 27mmHg), but a second surgical procedure or prolonged use of topical medication was not required. The total amount of endothelial cell loss, which was considered cumulatively at consecutive intervals throughout five years, was 7.7%. There was a tendency toward decreased phakic IOL anterior vault over time. No vision-threatening complications occurred [6].
Shimizu et al. assessed the early clinical outcomes of implanting a posterior chamber phakic IOL with a central hole (Visian V4 ICL; STAAR Surgical Company) for the correction of moderate to high myopia. The study included 20 eyes of 20 patients with spherical equivalents of -7.36 ± 2.13D. Before implantation of the IOL and at one week and one, three and six months postoperatively, the investigators assessed the safety, efficacy, predictability, stability and adverse events of the surgery. At six months, 95% and 100% eyes were within ±0.50D and ±1.00D of the targeted correction, respectively. The change in manifest refraction between the first week and six-month follow-up was 0.06±0.28D. A significant rise in IOP (including pupillary block) or a secondary cataract did not occur in any of the eyes during the period of observation [7].
LASIK and surface ablation
Alió et al. evaluated the clinical outcomes of LASIK in eyes with high myopia using optimised aspherical profiles and the 500-Hz Amaris excimer laser. The investigators used the 60-kHz IntraLase femtosecond laser (Abbott Medical Optics Inc.) to create the flap. The retrospective study included 51 eyes of 32 patients with high levels of myopia or myopic astigmatism (spherical equivalent ≥ 8.50D). Alió and colleagues recorded postoperative changes in visual acuity and refraction for six months. At three months postoperatively, a significant improvement (15 logMAR lines) was observed in distance UCVA but no significant changes were observed in the last three months of follow-up. Distance BCVA remained unchanged or improved in 98% of eyes at three months postoperatively. At six months postoperatively, 84.3% of eyes had a spherical equivalent within ±0.50D of emmetropia. A limited but significant induction of primary spherical aberration and coma was also found and a surgical enhancement was required in four eyes (7.8%) [8].
Comparative studies
Hassaballa and Macky retrospectively compared the outcomes of the Artisan anterior chamber IOL and the Visian ICL in 68 highly myopic eyes of 34 patients. The investigators found that both lenses demonstrated comparable safety, predictability and efficacy at one-year postoperatively. The mean preoperative spherical equivalent was -12.89±3.78D for the Artisan group and -12.44±4.15D for ICL group. The mean postoperative distance UCVA was 0.39±0.13 logMAR and 0.41±0.15 logMAR (6/15 Snellen equivalent) for the Artisan and ICL groups, respectively. The mean postoperative spherical equivalent was -0.86±0.50D for the Artisan group and -0.63±0.38D for the ICL group. The mean postoperative distance BCVA was 0.36±0.12 logMAR (6/12-3 Snellen equivalent) and 0.31±0.12 logMAR (6/12 Snellen equivalent) for the Artisan and ICL groups, respectively [3].
Nanavaty and Daya compared RLE with phakic IOLs and concluded that phakic IOLs provide better visual outcomes for distance. They found that, when laser ablative surgery is not possible, phakic IOLs and additive procedures are a safe option in myopic eyes with a deep anterior chamber, whereas in hyperopic eyes, RLE may be a better option. According to the authors, surgeons should consider various factors when choosing between RLE and a phakic IOL, including age, axial length, type and magnitude of refractive error, anterior segment configuration, endothelial cell count, and the patient’s desire for presbyopic correction. They recommend that surgeons do not consider RLE for patients under the age of 50, except in high hyperopes (≥ 4.00D) and when the anterior chamber depth is shallow and thus unsuitable for a phakic IOL. For these eyes, the authors recommend an age threshold of 45 years [9].
According to Nanavaty and Daya, the primary advantages of phakic lenses are rapid visual recovery, reversibility and a broader range of treatable ametropia than with RLE, high rates of predictability, and stability with the preservation of accommodation. With RLE, the risks of retinal detachment, cystoid macular oedema, glare, haloes and posterior capsular opacification remain. Risks with phakic IOLs include pigment dispersion, cataract formation, glaucoma and inflammation [9].
"The investigators found that both lenses demonstrated comparable safety, predictability and efficacy at one-year postoperatively."
Shin et al. compared changes in higher order aberrations (HOAs) in 30 eyes implanted with the Visian ICL and 33 eyes that underwent wavefront-guided LASEK. All eyes were highly myopic and had a spherical equivalent of -6.00D to -9.00D. At three months postoperatively, ICL implantation induced fewer ocular and corneal HOAs and resulted in better contrast sensitivity at mesopic levels compared with wavefront-guided LASEK. In the ICL group, HOAs changed for the entire ocular trefoil-y, spherical aberration, internal optic spherical aberration, and corneal trefoil-y. In the LASEK group, increased HOAs were observed for total HOAs, entire ocular and corneal spherical aberration, secondary astigmatism and tetrafoil. The ICL group had lower induced aberration values of entire ocular and corneal HOAs compared with the LASEK group. No significant differences in contrast sensitivity between groups at the photopic level were noted, but contrast sensitivity values were significantly lower for three and six cycles per degree in the LASEK group at the mesopic level. At the mesopic level, total HOAs, trefoil-y, spherical aberration and secondary astigmatism were higher in the LASEK group. A limitation of this study, however, was the relatively short follow-up period of three months [10].
Barsam and Allan conducted a meta-analysis of randomised controlled trials (RCTs) comparing refractive surgery with the excimer laser and phakic IOLs for the correction of between 6.00D to 20.00D of myopia. The investigators performed a comprehensive literature search using the Cochrane Collaboration methodology to identify RCTs and analysed data for efficacy outcomes, accuracy outcomes, safety outcomes, adverse effects and quality-of-life measures. The review included three RCTs and 228 eyes. Eyes in the phakic IOL group were less likely to lose two or more lines of BCVA at 12 months. Phakic IOL surgery scored more highly among patients on satisfaction and preference questionnaires [11,12].
The results of the meta-analysis showed that phakic IOLs are safer than excimer laser for the correction of high myopia one year postoperatively. Barsam and Allan noted, however, that some potential long-term risks that are unique to patients with phakic IOLs, such as continued endothelial cell loss and cataract formation, are not apparent within one-year of follow-up. Longer follow-up is required for a balanced evaluation of safety and to establish the ideal myopic range for excimer laser and phakic IOL treatments [11,12].

Case study
A 35-year-old doctor attended for consultation with the author to assess his suitability for safe refractive surgery. He was a soft contact lens wearer with no particular intolerance issues but wanted to explore options for contact lens independence. His quality of vision with glasses was poor. On examination his manifest dry refraction was:
R -10.50/-0.50 x127 6/7.5
L -7.75/-1.50 x 175 6/6-2
His ocular surface was in good condition (he had refrained from contact lens wear for one week prior to the consultation) with tear break-up times of nine seconds in each eye. He had 8mm pupils in mesopic conditions. Further assessment including cycloplegic refraction and dilated retinal examination was unremarkable. Elevation based corneal topography showed the thinnest corneal pachymetry readings to be 503 microns in the right eye and 517 microns in the left eye. Anterior chamber depth was 3.59mm in the right eye and 3.57mm in the left eye. After a discussion of what was involved as well as the risks and benefits he elected to have left thin flap advanced Z-LASIK with a 6mm optical zone followed later that day by a right implantable contact lens (ICL) under topical anaesthesia. At the day one postoperative visit his UCVA was 6/6-1 in each eye. His postoperative course was completely uneventful and at the 18-month postoperative visit unaided vision was 6/4 in each eye comfortably. Interestingly he does not notice any difference in the quality of vision between his two eyes.
Learning points from the case:
-
There is no absolute cut off for which ICL is preferred over minimally invasive advanced LASIK Most surgeons prefer to leave a residual stromal bed of over 300 microns under the LASIK flap in order to reduce the risk of post-LASIK ectasia in these larger ablations. For this reason the author felt that LASIK would be unsafe in his right eye but an acceptable approach for his left eye.
-
Many people worry unnecessarily about refractive surgery in patients with large pupils. While it is a consideration, with modern aspheric ablation profiles the author does not find that it interferes with quality of vision to a significant degree.
-
For large laser treatments it is preferable to use LASIK as opposed to surface treatments such as LASEK and PRK. The reason for this is that haze and refractive predictability become more problematic when more laser is being used.
-
It is an advantage to be able to offer patients more than just one modality of surgical treatment, as
refractive surgery cannot be safely or appropriately carried out with a ‘one size fits all’ approach.
References
1. Kempen JH, Mitchell P, Lee KE, et al. The prevalence of refractive errors among adults in the United States, Western Europe, and Australia. Archives of Ophthalmology 2004;122(4):495-505.
2. Buratto L, Ferrari M. Photorefractive keratectomy for myopia from 6.00 D to 10.00 D. Refract Corneal Surg 1993;9(2 Suppl):S34-6.
3. Hassaballa MA, Macky TA. Phakic intraocular lenses outcomes and complications: Artisan vs Visian ICL. Eye (Lond) 2011;25(10):1365-70.
4. Akcay L, Eser I, Kaplan AT, et al. Phakic anterior chamber lenses in very high myopia: an eighteen-month follow-up. Clin Experiment Ophthalmol 2012;40(3):275-81.
5. Lane SS, Waycaster C. Correction of high myopia with a phakic intraocular lens: interim analysis of clinical and patient-reported outcomes. J Cataract Refract Surg 2011;37(8):1426-33.
6. Alfonso JF, Baamonde B, Fernández-Vega L, et al. Posterior chamber collagen copolymer phakic intraocular lenses to correct myopia: five-year follow-up. J Cataract Refract Surg 2011;37(5):873-80.
7. Shimizu K, Kamiya K, Igarashi A, et al. Early clinical outcomes of implantation of posterior chamber phakic intraocular lens with a central hole (Hole ICL) for moderate to high myopia. Br J Ophthalmol 2012;96(3):409-12.
8. Alió JL, Vega-Estrada A, Piñero DP. Laser-assisted in situ keratomileusis in high levels of myopia with the amaris excimer laser using optimized aspherical profiles. Am J Ophthalmol 2011;152(6):954-63.e1.
9. Nanavaty MA, Daya SM. Refractive lens exchange versus phakic intraocular lenses. Curr Opin Ophthalmol 2012;23(1):54-61.
10. Shin JY, Ahn H, Seo KY, et al. Comparison of higher order aberrations after Implantable Collamer Lens implantation and wavefront-guided LASEK in high myopia. J Refract Surg 2012;28(2):106-111.
11. Barsam A, Allan BD. Excimer laser refractive surgery versus phakic intraocular lenses for the correction of moderate to high myopia. Cochrane Database Syst Rev 2014;6:CD007679.
12. Barsam A, Allan BD. Meta-analysis of randomized controlled trials comparing excimer laser and phakic intraocular lenses for myopia between 6.0 and 20.0 diopters. Cornea 2012;31(4):454-61.
Acknowledgement
Mr Barsam’s article originally appeared in the Journal of the Association of Optometrists: Optometry Today 2015;55(8):5-6. www.optometry.co.uk
Declaration of Competing Interests: None declared.
COMMENTS ARE WELCOME