Glaucoma is the most frequent cause of irreversible blindness worldwide and the second leading cause of blindness in the UK [1,2]. The global prevalence of glaucoma in 2010 was approximately 3.5% for people aged 40-80 years, according to Jonas et al. in a recent seminar feature on glaucoma in The Lancet [1].
Risk factors for both development and progression of glaucoma are older age, elevated intraocular pressure (IOP), sub-Saharan African ethnic origin, positive family history, stage of disease and high myopia [1]. Lowering of elevated IOP can reduce the risk of glaucoma progression [3]. Randomised trial data demonstrate that medical lowering of IOP preserves visual field in patients with open-angle glaucoma and progression may be halted if the IOP is lowered by 30-50% from baseline [1,4].
Managing and treating patients with glaucoma absorbs a considerable amount of ophthalmology hospital outpatient activity in the UK and refinement of the traditional glaucoma care pathway offers the twin benefit of more effective case-finding and enhanced capacity. The author outlines referral practice in Scotland, summarises updated National Institute for Health and Care Excellence (NICE) guideline recommendations from glaucoma referral, diagnosis and management, and considers emerging treatment perspectives.
Best practice in Scotland
Scotland has successfully promoted empowerment of optometrists for enhanced case-finding in glaucoma management and raised the threshold for secondary referral more than two years ago.
The Scottish Intercollegiate Guidelines Network (SIGN) national clinical guideline on glaucoma referral and safe discharge, issued in 2015, provides recommendations for best practice in the primary care assessment and referral of patients with suspected glaucoma into secondary eye care services and the safe discharge back into the community [5].
The guidance was designed to improve diagnostic accuracy and reduce inappropriate referrals to hospital eye services. Patients who have ocular hypertension (OHT) with IOP >25mmHg may be considered for referral to secondary eye care services irrespective of central corneal thickness (CCT). Patients who have OHT with IOP <26mmHg and CCT <555µm should be referred to secondary eye care services if they are aged ≤65. Patients who have OHT with IOP <26mmHg and CCT ≥555µm may be monitored in the community.
Irrespective of IOP, patients with one or more of the following findings should be referred to secondary eye care services:
- optic disc signs consistent with glaucoma in either eye
- a reproducible visual field defect consistent with glaucoma in either eye
- risk of angle closure (occludable angle):
- using Van Herick technique, if the peripheral anterior chamber width is one quarter or less of the corneal thickness
- using gonioscopy, if ≥270 degrees of posterior pigmented trabecular meshwork is not visible.
Community optometrists in Scotland often filter their own referrals and are resourced to do Goldmann applanation tonometry, disc images, corneal pachymetry, angle assessment and repeat visual field tests.
Updated glaucoma guideline from NICE raises national referral and treatment threshold in England
NICE published an updated guideline for diagnosing and managing chronic open-angle glaucoma (COAG) and OHT in November 2017 [2]. It replaces NICE Clinical Guideline (CG) 85 on glaucoma issued in 2009. The guideline scope has been extended to cover referral, with added or updated recommendations on testing and referral (case-finding), diagnosis, reassessment and treatment.
In areas where there is no glaucoma referral filtering scheme in place, around 30-40% of people referred to secondary eye care services are false positive referrals, according to NICE. It is now recommended that people with an IOP below 24mmHg should continue regular visits to their primary eye care professional (includes optometrists, GPs with a special interest in ophthalmology and community orthoptists) so any future increase in pressure is detected. The upper limit of statistically normal IOP is 21mmHg. There are over one million visits per year for glaucoma care in the NHS in England and it is estimated there are about 1.8 million people in the UK with an IOP of 22 or 23mmHg.
For OHT, knowledge of corneal thickness is no longer needed to decide whether or not to treat and a single IOP threshold of 24mmHg is now recommended for both onward referral and treatment. The independent guideline committee also made the recommendation to consider repeating measurements on another day before referral.
• Case-finding by primary eye care professionals
People planning and providing eye care services should use a service model that includes Goldmann-type applanation tonometry before referral for diagnosis of COAG and related conditions and should consider commissioning referral filtering services (e.g. repeated measures, enhanced case-finding or referral refinement) for COAG and related conditions.
Before referral for diagnosis of COAG and related conditions, all of the following tests are recommended: central visual field assessment using standard automated perimetry (full threshold or supra-threshold), optic nerve head assessment and fundus examination using stereoscopic – lamp biomicroscopy and optical coherence tomography (OCT) or optic nerve head image if available, IOP measurement using Goldmann-type applanation tonometry, and peripheral anterior chamber configuration and depth assessments using gonioscopy or, if not available or the patient prefers, the van Herick test or OCT.
A decision to refer should not be made solely on IOP measurement using non-contact tonometry. After considering repeat measures, referral is recommended if there is optic nerve head damage, a visual field defect consistent with glaucoma, or the IOP is 24mmHg or more measured using Goldmann-type applanation tonometry.
• Diagnosis
Following referral, an early assessment appointment should be considered when there is clinical concern based on the information provided. At time of diagnosis of OHT, the risk of future visual impairment should be assessed, taking account of IOP level, CCT, family history and life expectancy.
It was recommended in 2009 that an optic nerve head image is obtained at diagnosis for baseline documentation. The latest guideline clarifies that this may be acquired by a stereoscopic optic nerve head image (by biomicroscopy slit-lamp examination or stereo photography) or OCT, whichever is more readily available.
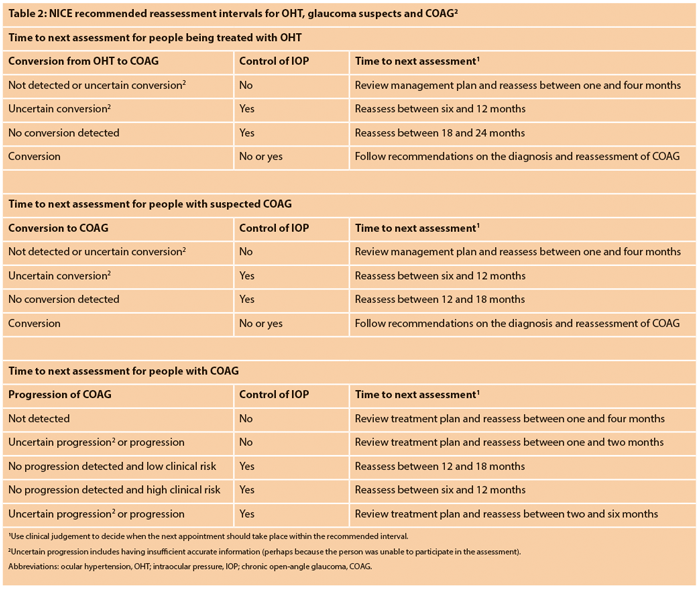
• Reassessment
At each assessment of people with COAG, suspected glaucoma or OHT, Goldmann applanation tonometry (slit-lamp mounted) and anterior segment slit-lamp examination with van Herick peripheral anterior chamber depth assessment when clinically indicated should be offered. When clinically indicated, repeat gonioscopy, for example, where a previous examination has been inconclusive or where there is a suspicion of a change in clinical status of the anterior chamber angle.
When clinically indicated, visual field testing using standard automated perimetry (central thresholding test) should be repeated for people with COAG and those suspected of having visual field defects who are being investigated for possible COAG. For people with OHT and those suspected of having COAG whose visual fields have previously been documented by standard threshold automated perimetry (central thresholding test) as being normal, repeat visual field testing using either a central thresholding test if available or a supra-threshold test is recommended.
The diagnosis algorithm and reassessment intervals are outlined in Table 1 and Table 2, respectively.
Table 1: NICE 2017 Glaucoma Guideline: Diagnosis algorithm.
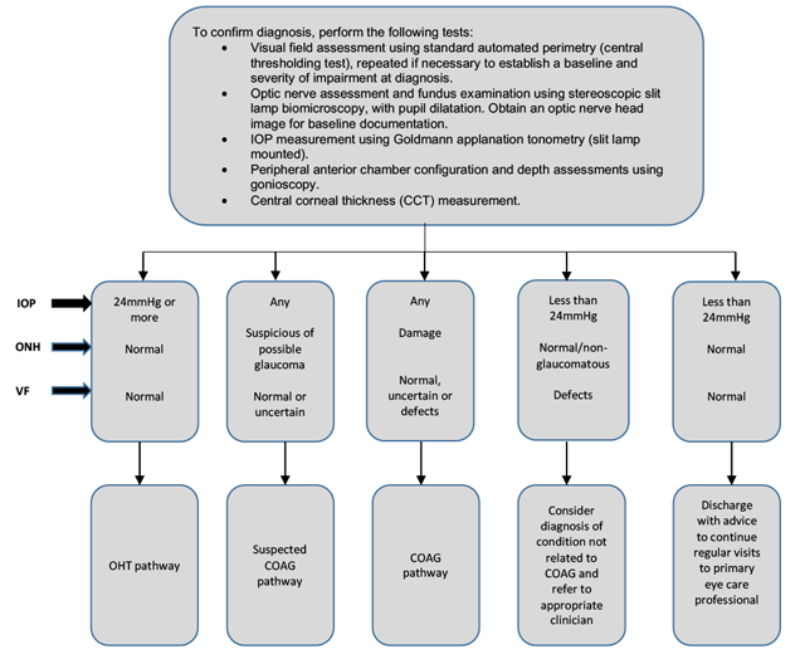
Abbreviations: intraocular pressure, IOP; optic nerve head, ONH; visual field, VF;
ocular hypertension, OHT; chronic open-angle glaucoma, COAG.
Source: © NICE [2017] Glaucoma: diagnosis and management. NICE guideline [NG81]. Published 1 November 2017.
Available from: www.nice.org.uk/guidance/ng81 All rights reserved. Subject to Notice of rights.
• Treatment
Treatment of glaucoma remains focused on control of IOP, with intensity of treatment and ongoing management guided by disease severity and progression as shown by visual field change, morphological change in the optic disc, and the likelihood of progressive sight loss. Ongoing patient reassessment encourages flexible clinical judgement about the frequency of visits and treatment options, including stopping treatment when the perceived risk to a sighted lifetime is low.
For people with OHT (IOP of 24mmHg or more) at risk of visual impairment, clinicians should offer treatment with a generic prostaglandin analogue (PGA). For those OHT patients whose current treatment is not reducing IOP sufficiently to prevent the risk of progression to sight loss, a drug from another therapeutic class should be offered (e.g. beta-blocker, carbonic anhydrase inhibitor or sympathomimetic). A combination of topical drugs from different therapeutic classes may be needed to control IOP.
Treatment adherence may be significantly affected by preservative toxicity. Preservative-free antiglaucoma medications should be offered to people who have an allergy to preservatives or people with clinically significant and symptomatic ocular surface disease, but only if they are at high risk of conversion to COAG. High risk of conversion to COAG was previously defined as IOP more than 25mmHg and up to 32mmHg and CCT less than 555µm, or IOP more than 32mmHg. These parameters have been removed from the latest guideline recommendations.
Treatment with a generic PGA should be offered to people with suspected COAG and IOP of 24mmHg or more, in line the recommendations on treatment for OHT.
The guideline clarifies that when switching to another monotherapy, the drug should be from another therapeutic class. This was considered important as committee members were aware of inappropriate switching of drugs from the same class, e.g. multiple PGA switches.
Generic PGAs are recommended as first-line treatment for people with COAG. Surgery with pharmacological augmentation (antimetabolite drug MMC) as indicated is recommended for people with advanced COAG, together with interim treatment using a generic PGA for those listed for trabeculectomy. Figure 1 summarises the recommended treatment algorithm for COAG.
Figure 1: Glaucoma guideline summary: chronic open-angle glaucoma algorithm*
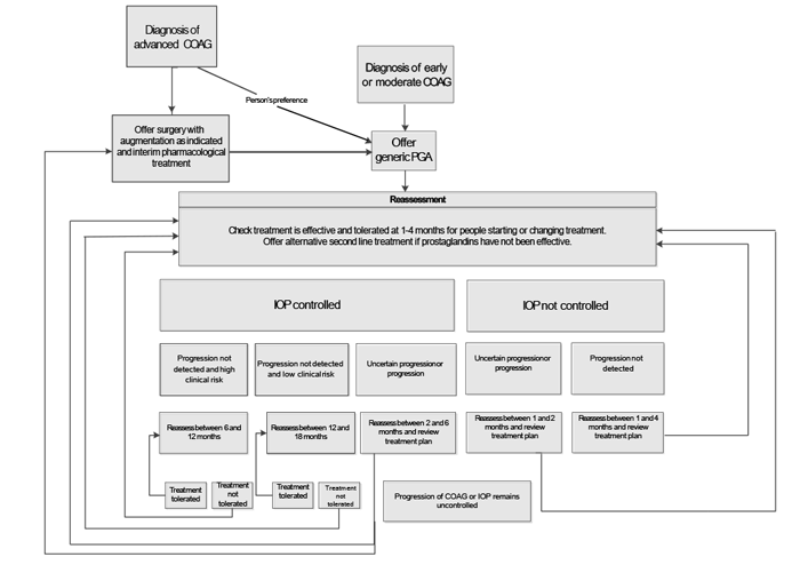
*At the time of publication (November 2017), not all generic PGAs had a UK marketing authorisation for first-line treatment. The prescriber should follow relevant professional guidance, taking full responsibility for the decision. Informed consent should be obtained and documented. See the General Medical Council’s ‘Prescribing guidance: prescribing unlicensed medicines’ for further information.
Abbreviations: chronic open-angle glaucoma, COAG; prostaglandin analogue, PGA; intraocular pressure, IOP.
Source: © NICE [2017] Glaucoma: diagnosis and management. NICE guideline [NG81]. Published 1 November 2017. Available from: www.nice.org.uk/guidance/ng81 All rights reserved. Subject to Notice of rights.
For COAG patients who cannot tolerate a prescribed treatment, clinicians are advised to consider a drug from another therapeutic class or preservative-free medications if there is evidence that the person is allergic to the preservative or has clinically significant and symptomatic ocular surface disease.
In people with COAG whose IOP has not been reduced sufficiently to prevent the risk of progression to sight loss despite pharmacological treatment, adherence and eye drop instillation technique should be checked. If satisfactory, one of the following options may be considered: drug treatment from another therapeutic class or combinations; laser trabeculoplasty; or surgery with pharmacological augmentation as indicated. If the drug treatment option is chosen, surgery or laser trabeculoplasty may be offered after trying drugs from two therapeutic classes.
People with suspected COAG and IOP less than 24mmHg should not be offered treatment but should be advised to continue regular visits to their primary eye care professional, at clinically appropriate intervals.
Stopping treatment for those with OHT or suspected COAG may be considered when there is both an acceptable IOP level and a low risk of ever developing visual impairment. When a patient opts to stop treatment after an informed discussion of benefits and risks, then IOP assessment in one to four months should be offered, with further reassessment if clinically indicated.
• Organisation of care
People with OHT, suspected COAG or COAG should have monitoring and treatment from a trained healthcare professional who has a specialist qualification, relevant experience and ability to detect a change in clinical status. Healthcare professionals should offer people the opportunity to discuss their diagnosis, referral, prognosis, treatment and discharge, and provide them with relevant information in an accessible format at initial and subsequent visits.
Discharge back to primary eye care services is recommended if people were referred for OHT but do not need treatment or were referred for suspected COAG but this is no longer suspected. A discharge summary should be given to people who have been assessed and discharged to primary care, together with a copy to their GP and, with patient consent, a copy of the relevant information sent to the primary eye care professional nominated by the patient. Patients should be advised to take their discharge summary with them when attending future sight tests.
Strengthened role of primary eye care professional welcomed
By raising the threshold for referral and treatment to an IOP of >24mmHg instead of >21mmHg, the latest glaucoma guideline recommendations from NICE are expected to dramatically decrease rates of false positive referrals to the hospital eye service in England. The guideline committee noted that there is no good quality evidence that people with IOP <24mmHg are at an increased risk of developing COAG.
Greater capacity in the hospital eye service will mean better care for those confirmed as being at increased risk of visual impairment. The recommendations are expected also to lead to improved accuracy of assessment prior to hospital referral and avert unnecessary appointments for patients who do not have glaucoma. Expansion and strengthening of the role of primary eye care professionals is broadly welcomed.
Emerging surgical options for mild to moderate glaucoma
Pressure-lowering treatment options for glaucoma include topical drugs, laser therapy and surgical intervention if other therapeutic interventions fail to prevent progression or there is advanced visual field loss at glaucoma diagnosis. The Treatment of Advanced Glaucoma Study is the first multicentre randomised controlled trial that will evaluate the comparative effectiveness of primary medical treatment against primary (augmented) trabeculectomy at two years for people with newly diagnosed advanced glaucoma [6]. Further follow-up evaluation through five years is being considered [6]. In another ongoing prospective multicentre randomised control trial (RCT), the LiGHT Trial Study Group will evaluate whether initial treatment with selective laser trabeculoplasty (SLT) is superior to initial treatment with topical medication for primary open-angle glaucoma (POAG) or OHT [7].
The scope of the updated NICE guideline did not include microinvasive glaucoma surgery (MIGS) devices, as the surveillance decision report identified that there was insufficient new evidence available to include surgical treatment options in the update of CG85, but surgical treatments will be considered at the next surveillance review of the guideline.
MIGS devices can be implanted in ab interno procedures to improve physiological aqueous outflow, significantly lower IOP and reduce the need for multiple topical medications. MIGS has fewer side-effects but lower efficacy compared with standard trabeculectomy and is an option for patients with mild to moderate POAG [1]. Minimally invasive stents can effectively lower IOP in combination with cataract surgery or as a stand-alone procedure and may reduce the need for multiple medical therapy in some patients.
“Minimally invasive stents can effectively lower IOP in combination with cataract surgery or as a stand-alone procedure.”
Tan and colleagues at Manchester Royal Eye Hospital reported 12-month follow-up results in a cohort of patients (n=39 eyes) who underwent XEN-45 (Allergan) implantation for glaucoma [8]. XEN-45 implant was shown to be an effective glaucoma treatment: the mean preoperative IOP was 24.9±7.8mmHg on three topical medications, which reduced to 14.5±3.4mmHg at month 12 (P<0.005) on 0.7 drops (P<0.005) [8].
Cypass micro-stent (Alcon) is inserted into the supraciliary space, enabling aqueous drainage through the uveoscleral pathway. At two-years in the COMPASS trial of patients with open-angle glaucoma undergoing cataract surgery, the proportion of eyes with ≥20% reduction from baseline in unmedicated diurnal IOP was 77% in the Cypass micro-stent in combination with cataract surgery group and 60% in the cataract surgery only group [9]. The proportion of patients medication free was markedly higher in the Cypass combination arm than in the cataract surgery only group (85% vs. 59% at 24 months). As a stand-alone procedure without lens surgery, Cypass micro-stent implantation effectively lowered IOP where prior medical treatments have failed in a multicentre, single-arm interventional study, with IOP reduced by 34.7% from baseline to one year [10].
Advocates believe that MIGS procedures hold considerable potential as part of a stepwise approach for mild to moderate glaucoma [11]. In a review of MIGS devices for glaucoma, Pillunat et al. noted that the different MIGS devices lower IOP between 3.2 and 12.6mmHg, with IOP reduction larger in patients with higher baseline IOP levels [8]. Also, the magnitude of the IOP lowering effects seems to be related to the number of shunts or stents implanted [12].
References
1. Jonas JB, Aung T, Bourne RR, et al. Glaucoma. Lancet 2017;390(10108):2183-93.
2. National Institute for Health and Care Excellence. Glaucoma: diagnosis and management. NICE guideline [NG81]. November 2017.
http://www.nice.org.uk/guidance/ng81
3. Heijl A, Leske MC, Bengtsson B, et al; Early Manifest Glaucoma Trial Group. Reduction of intraocular pressure and glaucoma progression: results from the Early Manifest Glaucoma Trial. Arch Ophthalmol 2002;120(10):1268-79.
4. Garway-Heath DF, Crabb DP, Bunce C, et al. Latanoprost for open-angle glaucoma (UKGTS): a randomised, multicentre, placebo-controlled trial. Lancet 2015;385:1295-304.
5. Scottish Intercollegiate Guidelines Network (SIGN). Glaucoma referral and safe discharge. Edinburgh: SIGN; 2015. (SIGN publication no. 144). [March 2015].
http://www.sign.ac.uk
6. King AJ, Fernie G, Azuara-Blanco A, et al. Treatment of Advanced Glaucoma Study: a multicentre randomised controlled trial comparing primary medical treatment with primary trabeculectomy for people with newly diagnosed advanced glaucoma-study protocol. Br J Ophthalmol 2017 Oct [Epub ahead of print].
7. Gazzard G, Konstantakopoulou E, Garway-Heath D, et al; LiGHT Trial Study Group. Laser in Glaucoma and Ocular Hypertension (LiGHT) Trial. A multicentre, randomised controlled trial: design and methodology. Br J Ophthalmol 2017 Sep [Epub ahead of print].
8. Tan SZ, Walkden A, Au L. One-year result of XEN45 implant for glaucoma: efficacy, safety, and postoperative management. Eye (Lond) 2017 Sep [Epub ahead of print].
9. Vold S, Ahmed IIK, Craven ER, et al; for the CyPass Study Group. Two-year COMPASS trial results: supraciliary microstenting with phacoemulsification in patients with open-angle glaucoma and cataracts. Ophthalmology 2016;123(10):2103-12.
10. García-Feijoo J, Rau M, Grisanti S, et al. Supraciliary micro-stent implantation for open-angle glaucoma failing topical therapy: 1 year results of a multicenter study. Am J Ophthalmol 2015;159(6):1075-81.
11. Gazzard G. Minimally invasive glaucoma surgery: MIGS. The Royal College of Ophthalmologists Focus Feature; Spring 2016.
12. Pillunat LE, Erb C, Jünemann AG, Kimmich F. Micro-invasive glaucoma surgery (MIGS): a review of surgical procedures using stents. Clin Ophthalmol 2017;11:1583-600.
Acknowledgment
Re Source for Table 1, Table 2 (adapted) and Figure 1
©NICE [2017] Glaucoma: diagnosis and management. NICE guideline [NG81]. Published 1 November 2017. Available from www.nice.org.uk/guidance/ng81 All rights reserved. Subject to Notice of rights.
NICE guidance is prepared for the National Health Service in England. All NICE guidance is subject to regular review and may be updated or withdrawn. NICE accepts no responsibility for the use of its content in this product/publication.
COMMENTS ARE WELCOME





