The point of this series is to find variations in practice and to raise them to you, the readership, so you can review and reflect upon them. I am aware there is not always a ‘correct’ way of doing things, however, we do have to try and act in an evidence-based manner and there are certain practices which can surely be standardised to some degree to prevent a postcode lottery in management.
The results from this survey highlight the issue of practice variance and the widely different things we as clinicians do.
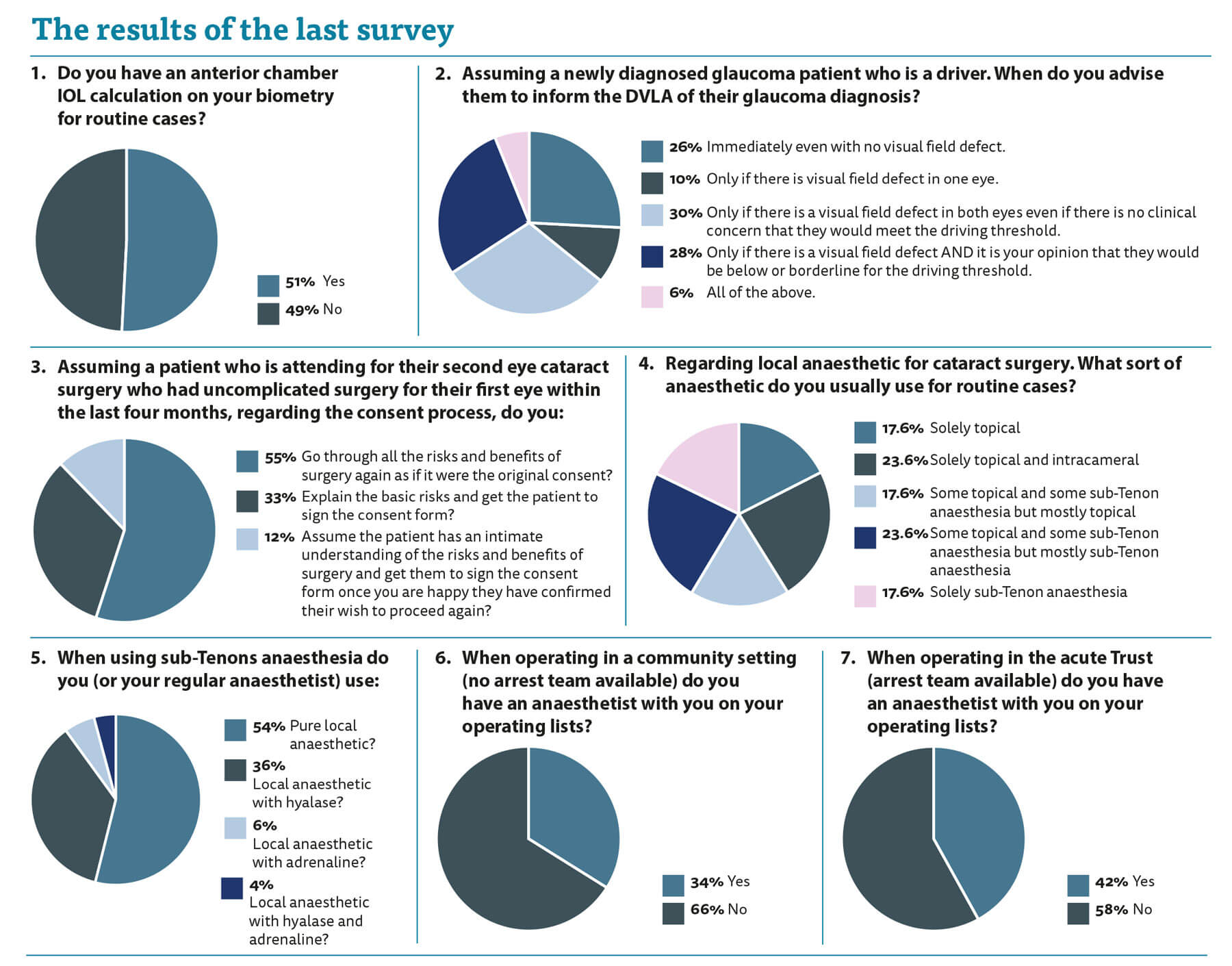
In almost a half of your practice the anterior chamber lens power is on the biometry. I think this is a mistake and I see regular cases where this IOL power has been picked for the posterior chamber lens. We all err but having that calculation on is tempting fate. If you pick the incorrect posterior chamber lens power then it is unlikely to be an issue. For example, if you picked a 22.5D IOL from the Bausch & Lomb (B&L) IOL calculation when you use an Alcon lens and you should have picked a 22.5D IOL or, at worst, a 22.0D IOL then very little harm is done. The patient can see and they will probably end up within 0.5D of emmetropia anyway.
Contrast that to picking the anterior chamber IOL power. Instead of the 22.5D IOL, which is the intended / ideal IOL, you choose an 18.5D IOL. Now the patient is grossly underpowered and has been left markedly hypermetropic. This makes for an unhappy patient and you are faced with exchanging the IOL, which then increases the risk of complications and a poor visual outcome.
In my view there is no need for the anterior chamber IOL calculation on the form as it is easily calculated if required. Not every unit has anterior chamber IOLs in stock so it is often utilised at another sitting after the aphakic eye has settled down. Even if it is immediately needed it is easy to calculate. If the A-constant of your routine lens is 119 and the A-constant of the anterior chamber IOL is 115 then you need to reduce the lens power by 4D. An aide memoire on the wall of your usual theatre can assist with these calculations.
The next question really highlights where we vary in practice. Almost a quarter of responders would ask the patient to refer themselves to the Driving and Vehicle Licensing Agency (DVLA) even if there were no field defect. I personally struggle to see the rationale behind this. We know that they meet the DVLA threshold for driving and therefore is this not a waste of taxpayers’ money and patient time? My personal practice is that this issue is assessed at each visit in every patient with bilateral visual field defects. If there is a concern that they may be borderline for reaching the threshold I ask them to self-refer to the DVLA for formal assessment. Having liaised with colleagues more intimately aware of this they pointed me to a RCOphth Focus article dated Summer 2013 which stated that the DVLA needs to be informed whenever there is a bilateral visual field defect. I remain sceptical at the necessity for this in a patient with a small arcuate in one eye and a tiny nasal step in the other, however, the primary point of this series is to highlight issues such as this and now I am appropriately educated, I will alter my practice.
Consent is a big issue we have discussed before and will continue to be a big issue. Second eye cataract surgery raises a significant consent issue. Patients are frequently brought back for second eye surgery without being seen in a clinic again and without having gone through the consent process. They are often asked to sign another consent form on the day. They have no time to consider their options as they are signing immediately before the surgery? Does it matter? They have already had one eye done, they were listed for the second eye and willingly came to the hospital for the procedure? But what if it goes wrong and they get a complication? Are we confident that they had true informed consent regarding the risks, or do we fear that because their first eye went well, they are attending with the erroneous impression that their second eye will be fine and uncomplicated?
Regarding the mix of local anaesthetic techniques, we can see a reasonably even spread. It is interesting to see that more than half of you or your anaesthetists use pure local anaesthetic without an additive. Ten percent use adrenaline which I have concerns about. We know that the optic nerve head blood supply can be precarious particularly in glaucoma patients and we also know that the central retinal artery is an end-artery. Is it wise to use adrenaline around an end-artery?
Hyalase is used in approximately 40% of block anaesthetics. The aim is to improve spread of the anaesthetic and aid akinesis. Since the landmark study by Alwitry et al. [1] I have not used hyalase myself as it does not seem to have any significant effect on efficacy of block after five minutes of administration.
Regarding the presence of an anaesthetist for the operating list the results are self-explanatory. I have an anaesthetist to speed throughput of cases and also in case one of the patients goes off. I have had cases of severe bradycardia and hypoglycaemia when I was glad for the presence of an anaesthetic colleague to save the day and the patient. As always, this commentary is not designed to offer guidance but more to stimulate questions and reflection on practice, hopefully encouraging us to move towards consistent evidence-based practice.
Reference
1. Alwitry A, Chaudhary S, Gopee K, et al. Effect of hyaluronidase on ocular motility in sub-Tenon’s anesthesia: randomized controlled trial. J Cataract Refract Surg 2002;28(8):1420-3.
COMMENTS ARE WELCOME






