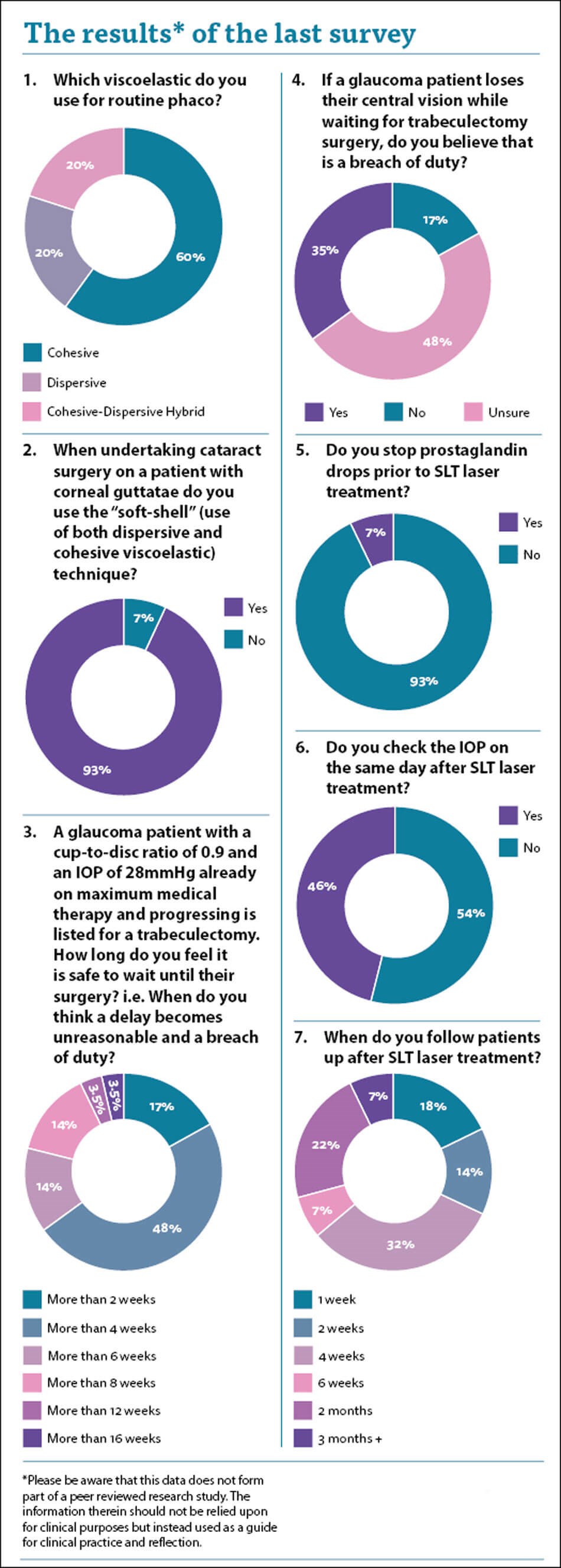
The first question really highlights how we can become enshrined in the way we do things. I genuinely thought that everyone was using a cohesive viscoelastic for their cataract surgery. It was what I was taught, and I believe everyone in my region uses a cohesive viscoelastic. As we know, viscoelastics, also referred to as ophthalmic viscosurgical devices (OVDs), are viscous substances that create space in the anterior chamber for us to work.
Dispersive OVDs have a syrupy consistency, and they are able to flow like thick liquids. This gives dispersive OVDs the ability to coat ocular structures well and protect the corneal endothelium, and this protective coating is not easily washed away by the flow of balanced salt solution into the eye and the suction / aspiration of the phacoemulsification probe. The downside is that they are harder to remove and require actively going around the anterior chamber to vacuum it up.
Cohesive OVDs are more solid in nature and they have the consistency of gelatin. Because they are much thicker, they are able to maintain space and pressurise the eye well. They are thus very good at keeping the anterior chamber formed, keeping the anterior capsule flat during capsulorrhexis creation, and opening the empty capsular bag open for IOL insertion.
There is a clear spread in practice. The majority of surgeons use a cohesive viscoelastic, while one in five use a hybrid cohesive-dispersive. I was surprised to see that one fifth of surgeons routinely use just a dispersive viscoelastic. I wonder whether this is good enough for all cases including those with a shallow anterior chamber. Clearly the endothelial protection will be good because the viscoelastic stays around, but what about the space-creation function of the OVD? Are we confident that these agents can keep the anterior chamber deep enough to avoid proximity related iatrogenic damage to ocular structures from instruments and ultrasound power?
This leads nicely on to the “soft-shell” technique which utilises both a layer / “shell” of dispersive viscoelastic adjacent to the corneal endothelium, and then a cohesive viscoelastic beneath it to create and maintain space. It is a technique I tend to use for all my patients where I have a concern about the potential of endothelial cell loss. I was pleased to see that 93% of respondents do the same. I am slightly concerned about the 7% who do not, and I am aware that some surgeons do not believe in it. With 93% of respondents, and also many of the experts who I deal with, of the opinion that it is optimal care then they may face criticism if corneas decompensate, and this technique was not utilised.
The next question is difficult and controversial. If my father was the patient described, then I would want him to have his glaucoma surgery within two weeks of the decision to operate. I know that his glaucoma is unstable, and he will be getting progressively worse. His glaucoma is already bad, and he is at risk of losing what little he has left. However, there are massive capacity issues across the whole NHS. We do not have enough glaucoma surgeons and we do not have enough theatre capacity to allow us to guarantee a glaucoma operation immediately, or almost immediately. We could argue that such a high-risk patient should take priority and that another patient should be cancelled, but then that other patient’s care is compromised. Furthermore, all the patients on the intended list may be equally in need of surgery.
Despite a wide variation in opinion, almost half of you felt that four weeks was the cut-off point where a delay led to avoidable potential harm. I think this is reasonable, but is it fair to criticise Trusts which cannot meet this arbitrary deadline when they do not have the staff or facilities to achieve it? I think not, but for that individual patient the consequences are dire, and I myself would not be happy if my father lost vision because of the delay. A third of you felt that if a patient lost vision while waiting for surgery it would be a breach of duty, while half of you were unsure. There is no clear answer, and the way I try to think of it is to assess whether there was an avoidable delay to surgery or whether the Trust did the best they could in their circumstances.
With the new National Institute of Health & Care Excellence (NICE) Glaucoma guidance recommending selective laser trabeculoplasty (SLT) laser for treatable glaucoma and ocular hypertension (OHT), I was interested to see whether there was consistency around the management of patients undergoing this laser.
I was told once that prostaglandins should be stopped prior to SLT laser, however I was never quite sure why, and it is not my current standard practice. Seven percent of respondents stop prostaglandin drops. There is some mild evidence to support this, but I was not convinced by it. Scherer described a difference in IOP reduction when comparing prostaglandin analogue users and nonusers when the baseline IOP was 17.75 mmHg [1]. The SLT efficacy decreases with lower IOP [2]. It is possible to speculate that with a lower baseline IOP, the effects of eye drops are more prominent than the effects of SLT and this can explain the reason why Scherer found a difference between the groups. In an article published in 2009, Alvarado et al. described in a translational model (in vitro-in vivo) a possible common action mechanism for prostaglandin analogues and SLT [2]. The authors concluded that prostaglandin analogues and SLT may share the same pathway (at the trabecular meshwork) for IOP reduction and therefore stopping prostaglandins would enhance SLT efficacy. There is also contrary evidence which contradicts this [3].
It was interesting to see an almost 50:50 split in those of you who check the patients’ IOP on the same day after their SLT. I have been doing so for some time and picked up no cases where their intraocular pressure has spiked and needed treatment. I was contemplating stopping the practice and am reassured to some degree that almost half of respondents do not check the IOP on the same day. Follow-up showed a massive variance with a large spread of practice. There is clearly no consensus and no correct follow-up time to follow. My personal practice is to bring these patients back in six weeks to determine efficacy and discuss reducing eye drops. It was interesting to note only 7% of you agreed with me. I may alter my practice to align with the 32% of you who bring patients back in four weeks.
References
1. Scherer WJ. Effect of topical prostaglandin analog use on outcome following selective laser trabeculoplasty. J Ocul Pharmacol Ther 2007;23(5):503-12.
2. Alvarado J, Iguchi R, Juster R, et al. From the bedside to the bench and back again: predicting and improving the outcomes of SLT glaucoma therapy. Trans Am Ophthalmol Soc 2009;107:167-81.
3. D Singh, Coote MA, O’Hare F, et al. Topical prostaglandin analogues do not affect selective laser trabeculoplasty outcomes. Eye (Lond) 2009;23(12):2194-9.
COMMENTS ARE WELCOME






